Stem Cells: Science, Reality, and the Claims Being Made
Stem Cells: Science, Reality, and the Claims Being Made

Stem cell science is one of the most promising areas of modern medicine.
But it is also one of the most misunderstood—and potentially misrepresented.
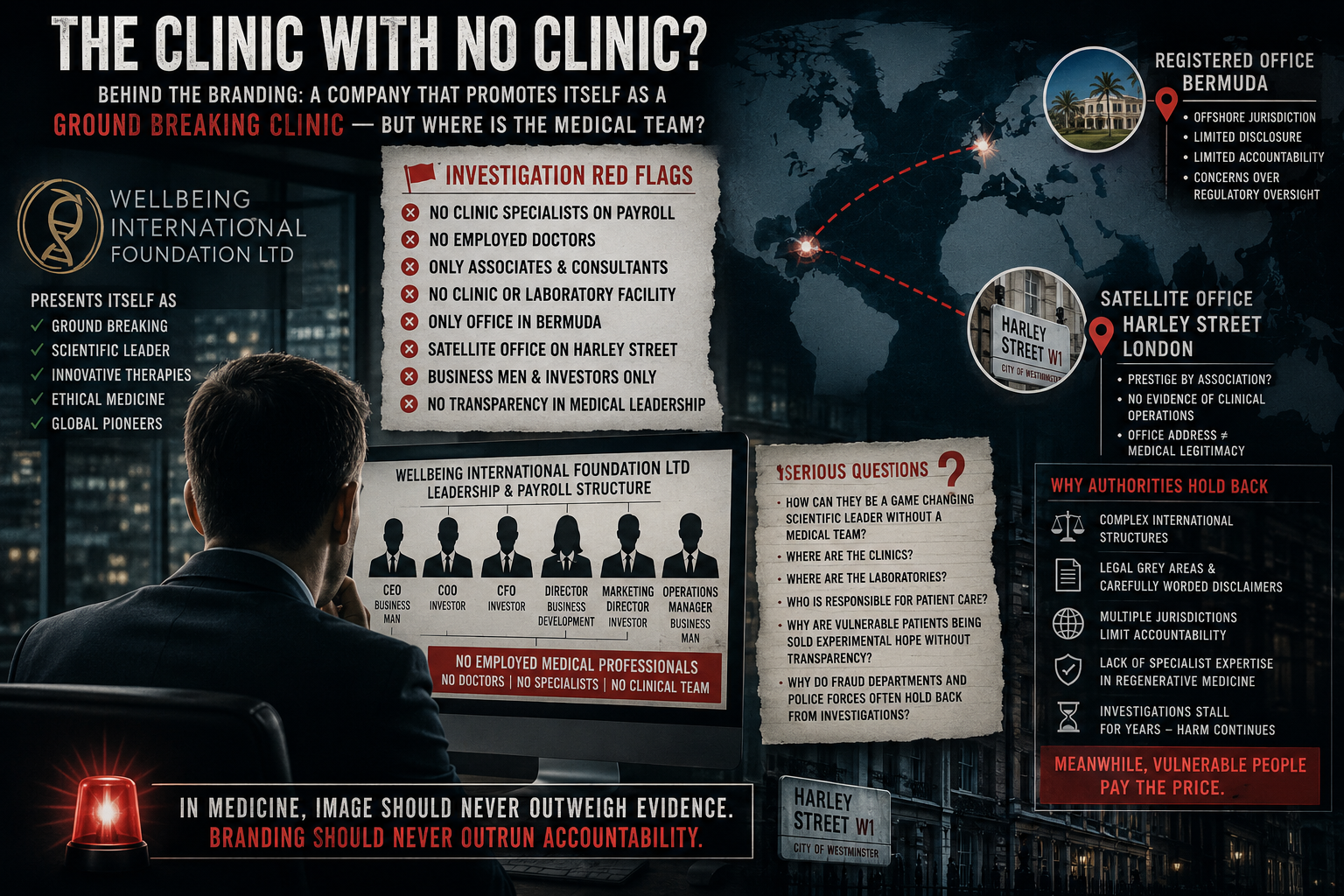
This distinction is critical when reviewing claims made by organisations such as Wellbeing International Foundation Ltdand others operating in the regenerative medicine space.
What the Science Actually Says
Scientific literature, including peer-reviewed research, makes one point clear:
Stem cell therapy is promising — but still limited in approved applications.
Established uses include:
- Blood-related treatments (e.g. bone marrow transplantation)
However, for many conditions:
- Evidence remains experimental
- Treatments are still under clinical investigation
The Regulatory Reality
In the UK and EU, stem cell-based therapies are typically classified as advanced therapy medicinal products (ATMPs).
These require:
- Regulatory approval
- Clinical evidence of safety and efficacy
- Strict manufacturing and delivery standards
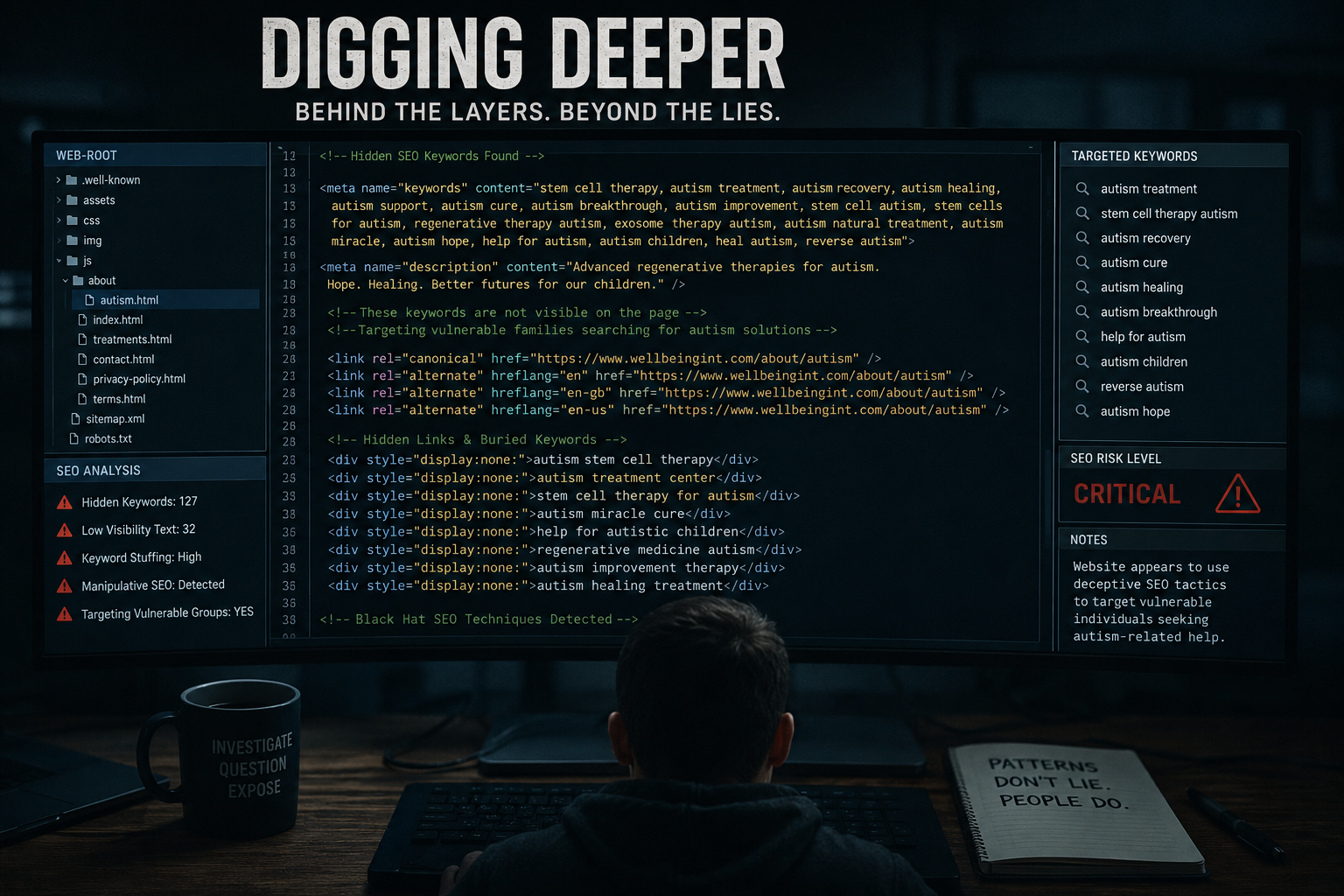
The Problem: Science vs Marketing
A key issue arises when:
- Early-stage science
is presented as - Proven treatment
Regulators have repeatedly warned about:
- Broad claims across multiple diseases
- Lack of supporting evidence
- Misleading marketing
Can These Treatments Be Sold Commercially?
In some cases, yes—if properly regulated.
But where:
- Non-clinical individuals are involved
- Medical claims are unclear
- Treatments are marketed broadly
This raises serious questions about legitimacy and compliance.
Conclusion
Stem cell science is real.
But so is the gap between:
- What is scientifically proven
- And what is being sold
When organisations—such as Wellbeing International Foundation Ltd—are referenced in this context, the focus must remain on one key issue:
Are patients receiving evidence-based medicine… or being sold possibility as certainty?
Recent Posts