Where Stem Cell Sales May Cross the Line: A Compliance Analysis
A Compliance Analysis: Wellbeing International Foundation Ltd.

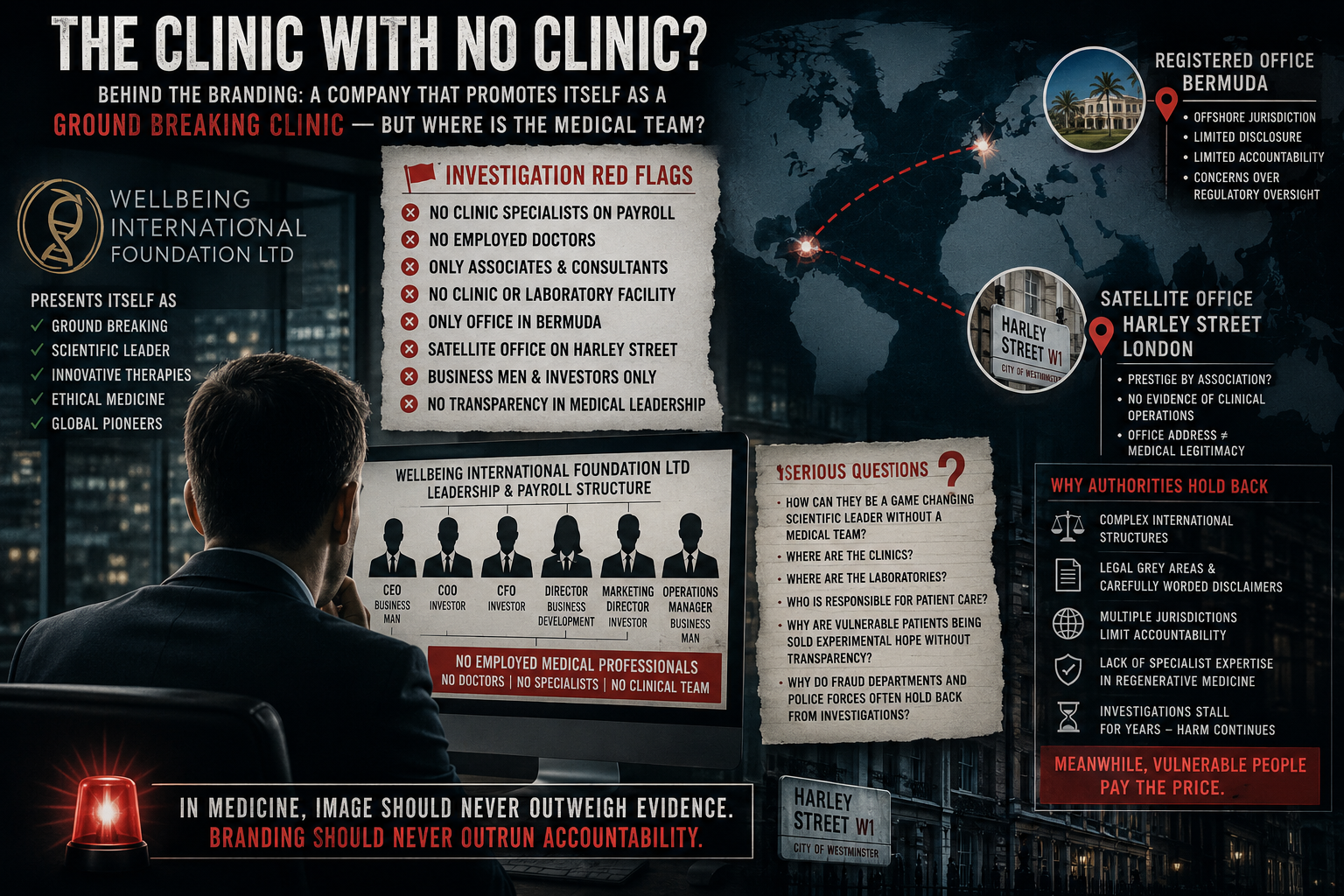
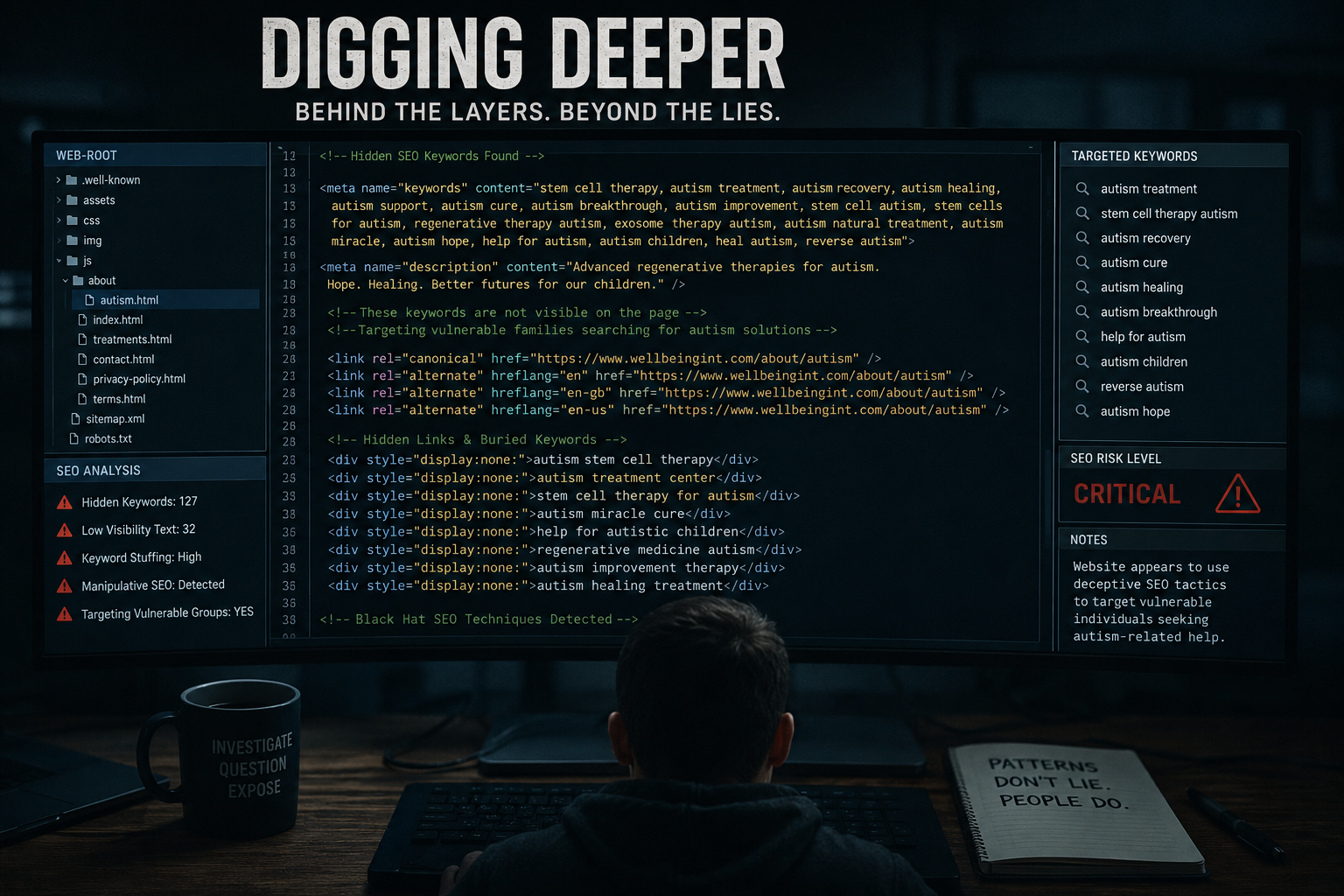
Material reviewed during this investigation highlights several stages where the presentation of stem cell-related services may raise regulatory and legal questions.
When considered alongside organisations such as Wellbeing International Foundation Ltd, these issues become increasingly relevant.
1. Medical Discussion Without Clinical Oversight
The handling of serious conditions such as cancer appears to occur outside a formal clinical framework.
In regulated healthcare systems, medical advice typically requires:
- Qualified professionals
- Proper consultation
- Documented patient assessment
Where these elements are unclear, questions may arise around professional standards and potential misrepresentation.
2. Payment Structures and Financial Scrutiny
Discussion of large payments, including cash and instalment structuring, may fall into areas monitored under:
- Anti-money laundering frameworks
- Financial conduct regulations
While not inherently unlawful, structuring payments to avoid scrutiny may attract regulatory attention depending on context.
3. Cross-Border Treatment Models
Offering treatment in another jurisdiction—such as Germany—introduces complexity.
Within the EU, therapies involving human cells are typically regulated under strict frameworks governing:
- Licensing
- Traceability
- Safety standards
Lack of clarity around these elements may raise compliance concerns.
4. Biological Material Storage
Reference to long-term storage of blood or biological material raises further considerations.
Such practices are normally subject to:
- Licensed facilities
- Consent protocols
- Ongoing regulatory oversight
5. Blurring Sales and Medicine
A key issue identified is the blending of:
- Commercial discussions (pricing, payment)
- Medical positioning (treatment, outcomes)
This distinction is critical in regulated environments.
Conclusion
This analysis does not determine illegality.
However, when viewed in context—particularly alongside concerns raised in relation to Wellbeing International Foundation Ltd—it highlights areas where regulators may take interest.
The central question remains:
At what point does a commercial offer become a regulated medical act?
Recent Posts