STEM CELL SCANDAL
FALSE HOPE. REAL HARM. MILLIONS LOST.

They promise miracles.
They sell “regeneration.”
They target the desperate.
And behind the glossy websites and white coats lies a booming global marketplace built on treatments that regulators repeatedly warn are unproven, unapproved — and sometimes dangerous.
This is the stem cell trap.
A BOOMING INDUSTRY BUILT ON HOPE
In 2016, researchers mapped the U.S. stem cell marketplace and found 351 businesses operating 570 clinics marketing stem cell interventions directly to consumers.
By March 2021, that number had exploded to 1,480 businesses running 2,754 clinics.
That’s not growth.
That’s an industry surge.
Source (2016 study):
https://www.sciencedirect.com/science/article/pii/S1934590916301576
Source (2021 expansion data):
https://pubmed.ncbi.nlm.nih.gov/34739831/
The majority of these clinics were advertising treatments for conditions for which no FDA approval existed.
The U.S. Food and Drug Administration has repeatedly warned consumers that many regenerative medicine products marketed directly to patients are not approved and require oversight in clinical trials.
FDA consumer warning:
https://www.fda.gov/vaccines-blood-biologics/consumers-biologics/important-patient-and-consumer-information-about-regenerative-medicine-therapies
WHO ARE THEY TARGETING?
The condition lists read like a catalogue of human suffering:
- Autism
- Alzheimer’s disease
- Muscular dystrophy
- Spinal cord injury
- Multiple sclerosis
- Stroke
- Parkinson’s
- Heart disease
- COPD
- Chronic pain
- And even cancer-related claims
Source (Pew Charitable Trusts):
https://www.pew.org/-/media/assets/2021/05/harms-linked-to-unapproved-stem-cell-interventions-highlight-need-for-greater-fda-enforcement.pdf
Source (market condition spread analysis):
https://multimedia.elsevier.es/PublicationsMultimediaV1/item/multimedia/X0213371717618287%3A277v52n196-90461828mmc1.pdf
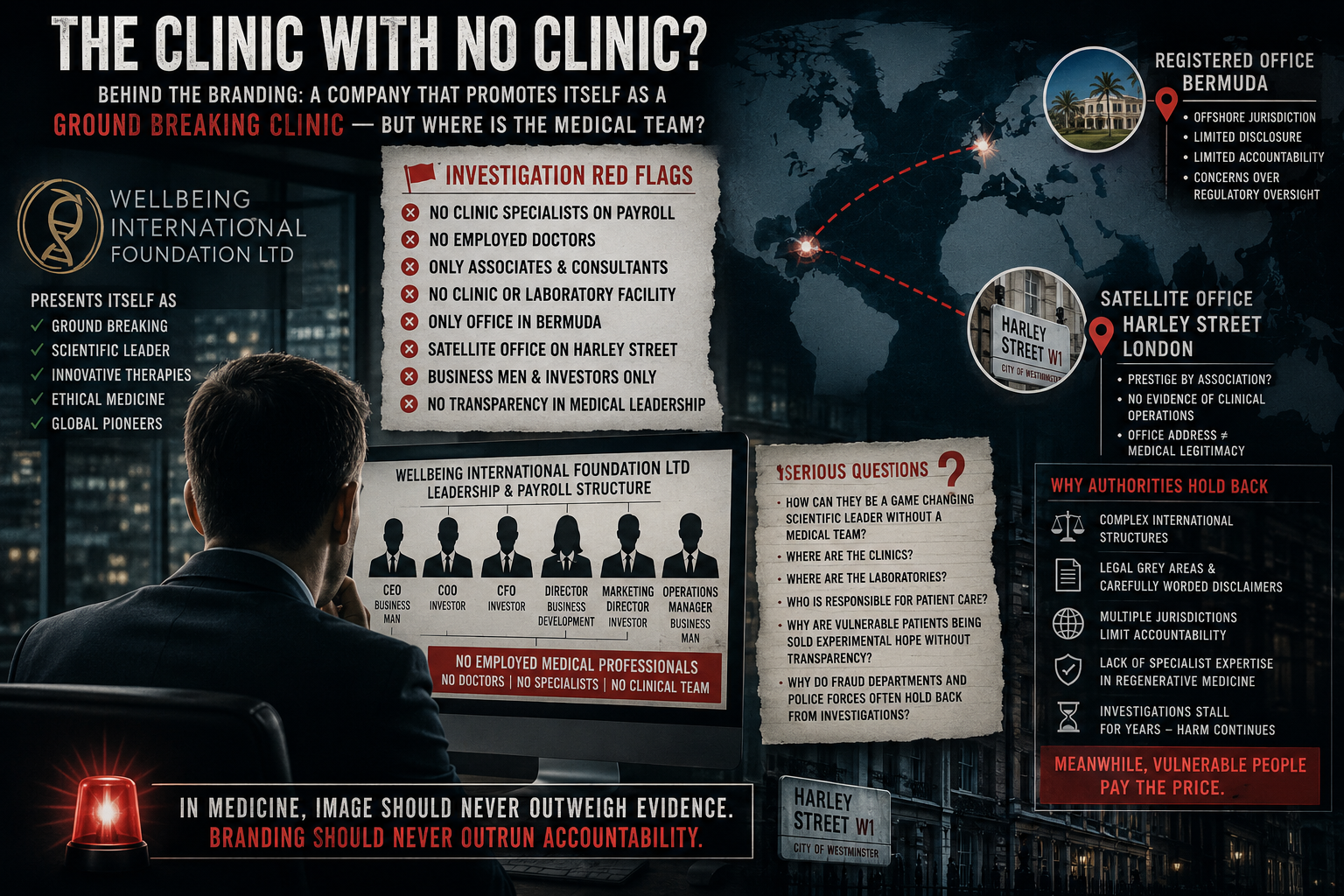
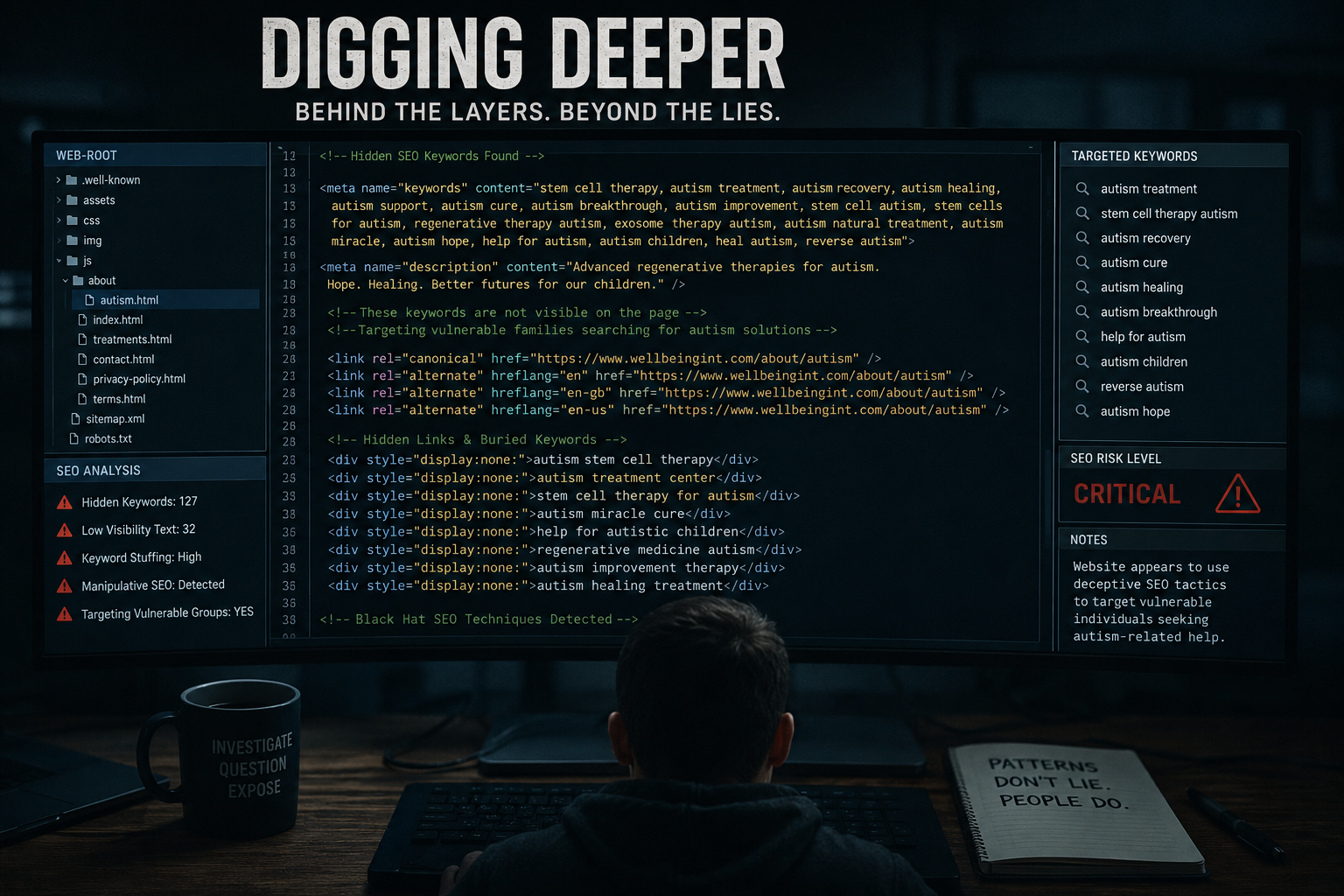
Autism treatments have been among the most controversial and emotionally charged marketing claims.
Federal investigations and reporting have documented clinics promoting stem cells as solutions for autism and blindness.
STAT News reporting:
https://www.statnews.com/2018/10/18/feds-crack-down-on-stem-cell-clinics-that-touted-autism-treatments-blindness-cures/
These are not minor ailments.
These are families looking for hope.
These are terminal patients looking for time.
THE HUMAN COST: BLINDNESS AND DOCUMENTED HARM
This is not theoretical.
In a landmark case published in the New England Journal of Medicine, patients suffered severe and permanent vision loss after receiving unproven stem cell injections in their eyes.
NEJM case report:
https://www.nejm.org/doi/full/10.1056/NEJMoa1609583
Three women were left blind or legally blind after the procedure, widely reported at the time.
Stanford Medicine coverage:
https://med.stanford.edu/news/all-news/2017/03/unproven-stem-cell-therapy-blinds-three-patients-at-florida.html
And that’s just one documented cluster.
A Pew Charitable Trusts review identified 360 people with reported adverse events linked to unapproved stem cell interventions between 2004 and September 2020.
Crucially, Pew noted most adverse events were not found in official reporting databases — meaning the real number could be higher.
This is the iceberg problem.
What you see reported is likely a fraction of what actually occurs.
FOLLOW THE MONEY
When regulators move in, the financial picture becomes clearer.
In one enforcement action involving Georgia consumers, authorities ordered:
- $3,310,146 in refunds
- 479 consumers refunded
- Treatments often costing around $5,000 per joint injection
Georgia Attorney General announcement:
https://law.georgia.gov/press-releases/2025-01-08/carr-superior-healthcare-co-founders-banned-marketing-stem-cell
That’s nearly 500 consumers in just one state case.
Now multiply that across thousands of clinics operating nationwide.
There is no global database tallying every financial victim. But enforcement actions show a clear pattern:
High fees.
Broad claims.
Vulnerable patients.
Refunds only after regulators intervene.
HOW THE SALES MACHINE WORKS
According to enforcement filings, clinics have used:
- Free seminars
- Social media advertising
- YouTube promotions
- Infomercial-style marketing
- Email campaigns
- Print ads
Source (Georgia case details):
https://law.georgia.gov/press-releases/2025-01-08/carr-superior-healthcare-co-founders-banned-marketing-stem-cell
This is consumer sales strategy — not specialist referral medicine.
Patients are often asked to sign waivers acknowledging risk while being told of potential life-changing benefits.
The FDA continues issuing warning letters to clinics marketing unapproved regenerative products.
FDA enforcement page:
https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/compliance-actions-and-activities/warning-letters
THE REGULATORY GAP
The core issue isn’t stem cell science itself.
Legitimate stem cell therapies do exist — but only for specific approved uses and under strict regulation.
The problem is the explosion of clinics marketing interventions outside those approvals, often framing them as cutting-edge breakthroughs.
When hope becomes product.
When desperation becomes revenue.
When medicine becomes marketing.
THE UNANSWERED QUESTION
There is no central number telling us exactly how many people worldwide have:
- Paid for unproven treatments
- Suffered adverse effects
- Been financially devastated
- Been left worse than before
But here’s what we do know:
✔ Thousands of clinics have operated in the U.S. alone
✔ Hundreds of documented injury cases exist
✔ Millions of dollars have been ordered refunded
✔ Regulators continue to intervene
And the marketplace continues.
WHAT PATIENTS SHOULD ASK
Before committing to any stem cell treatment outside a regulated clinical trial:
- Is this FDA-approved for my condition?
- Is this part of a registered, regulated clinical trial?
- Who oversees adverse event reporting?
- What peer-reviewed evidence supports this claim?
FDA guidance for consumers:
https://www.fda.gov/vaccines-blood-biologics/consumers-biologics/important-patient-and-consumer-information-about-regenerative-medicine-therapies
THE BOTTOM LINE
The stem cell promise is real in laboratories.
But in the commercial marketplace, the evidence shows:
- Rapid expansion.
- Broad unproven claims.
- Documented injuries.
- Documented financial losses.
Hope should never be a revenue model.
Recent Posts