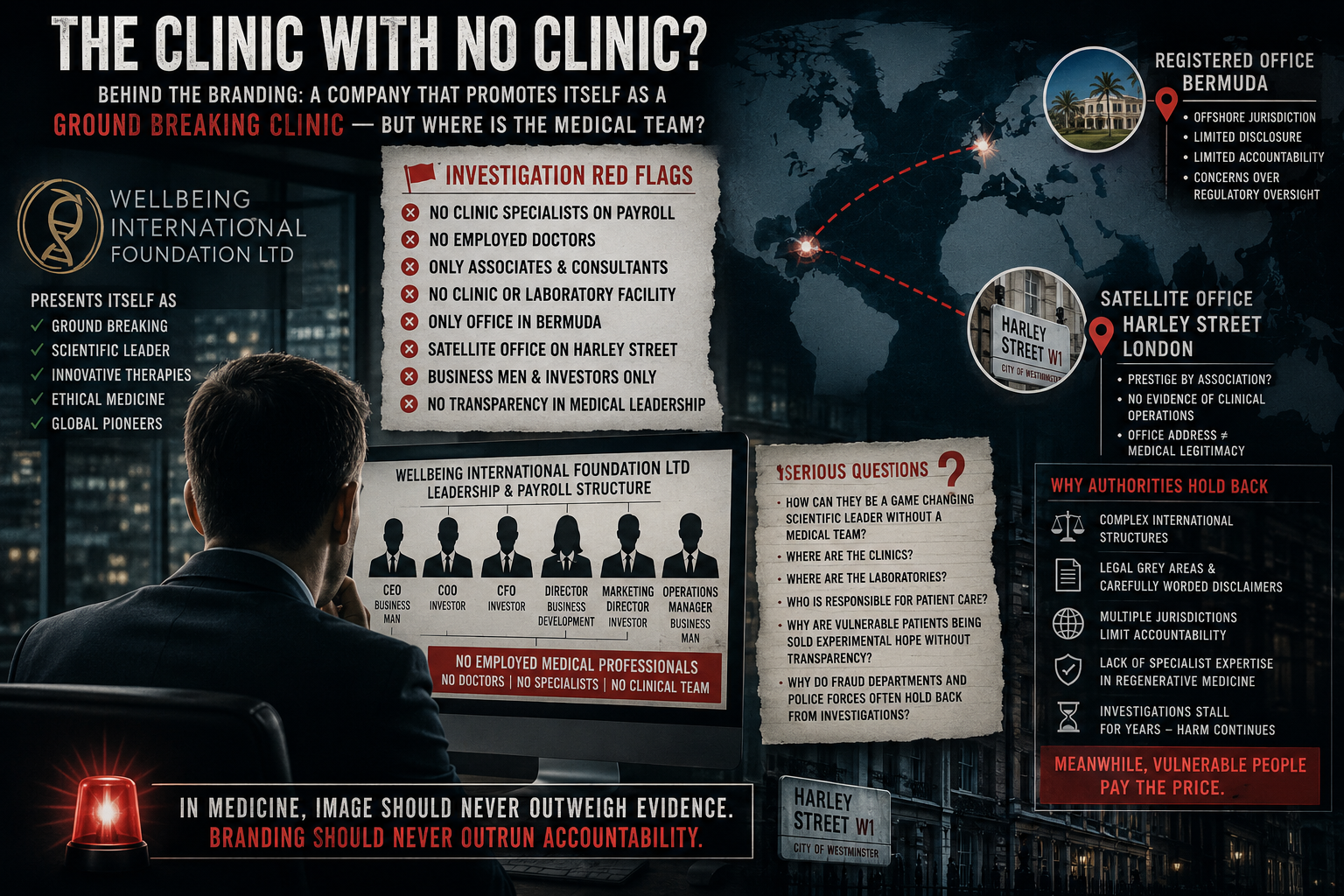
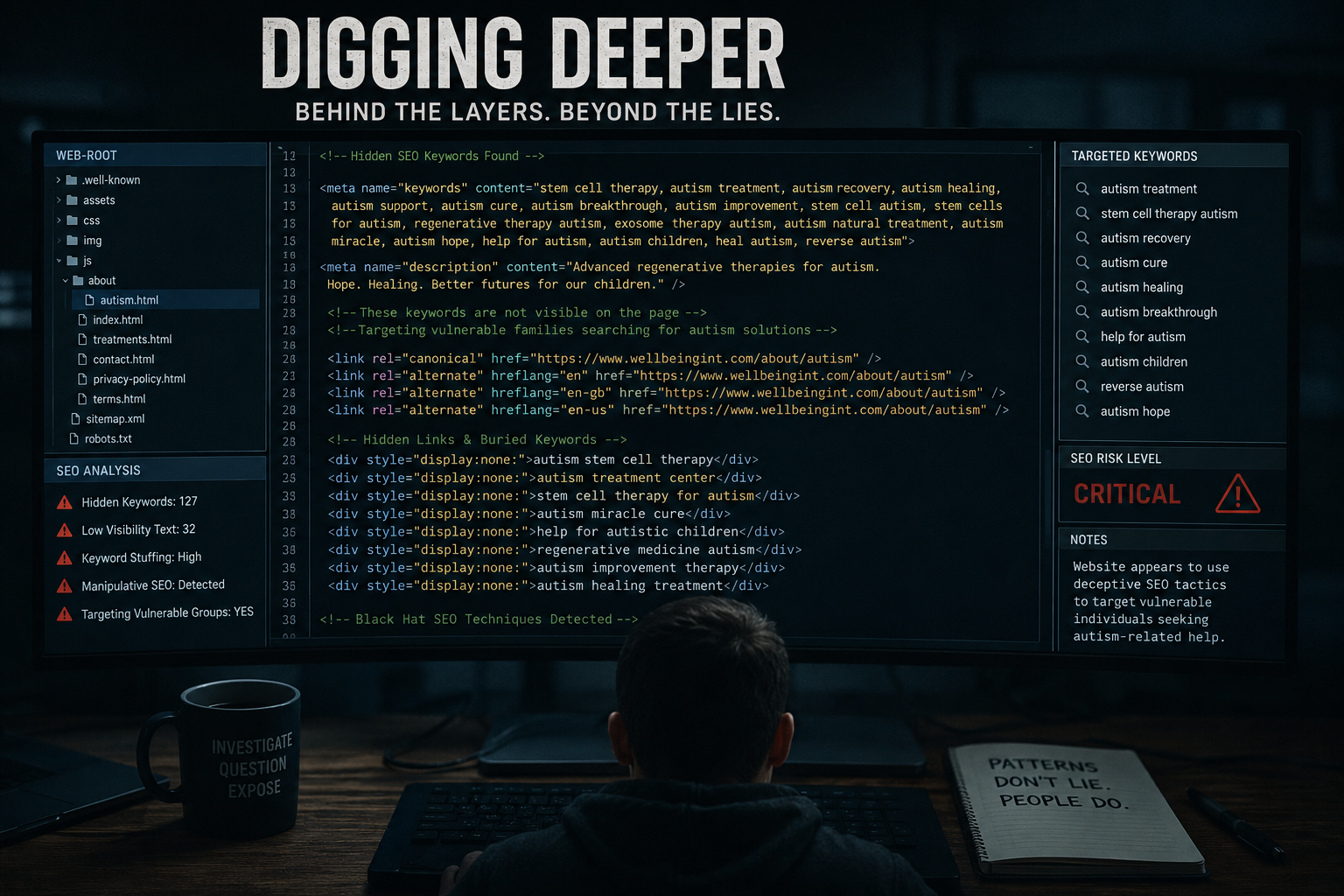
What Is Wellbeing International Foundation Ltd?
UK STEM CELL CLINICS UNDER THE MICROSCOPE

Questions Raised Over Private Regenerative Medicine Providers — Including Wellbeing International Foundation Ltd
By Steven Millard | Investigative Report
Britain’s stem cell marketplace is growing.
So are the questions.
Across the UK, private regenerative medicine providers are marketing stem cell interventions for a wide range of conditions — from chronic pain to neurological disorders — while regulators such as the Medicines and Healthcare products Regulatory Agency (MHRA) continue to warn that many stem cell products require strict licensing before being offered to patients.
At the heart of the issue is not stem cell science itself.
It is oversight.
And transparency.
And whether patients fully understand the regulatory status of what they are being offered.
The Regulatory Framework: What the MHRA Says
Under UK law, stem cell products that qualify as medicinal products — including most advanced cell therapies — must be authorised by the MHRA before being marketed.
These are classified as Advanced Therapy Medicinal Products (ATMPs).
MHRA guidance explains that any such product must undergo clinical trials and regulatory approval unless it falls under specific exemptions, such as the limited “hospital exemption” framework.
MHRA enforcement guidance:
https://www.gov.uk/government/publications/clinical-trials-regulations-enforcement-provisions/clinical-trials-regulations-enforcement-provisions
Regulatory overview of stem cell therapies in the UK:
https://www.eurostemcell.org/regulation-stem-cell-research-united-kingdom
The MHRA also retains powers to issue warning notices, compliance orders, and in rare cases pursue criminal enforcement where medicinal products are unlawfully marketed.
Parliamentary Concerns About Regulatory Gaps
Written evidence submitted to Parliament has warned that some stem cell interventions available in the UK may fall outside robust oversight frameworks.
Experts noted concerns including:
- Misleading marketing to vulnerable patients
- High costs associated with unproven therapies
- Ambiguity in classification between medical procedures and medicinal products
Parliamentary written evidence:
https://committees.parliament.uk/writtenevidence/111351/pdf/
The concern is not theoretical.
It is structural.
Where regulatory classification is unclear, enforcement can become reactive rather than proactive.
The Wider Marketplace
Investigations into private regenerative medicine providers — both in the UK and internationally — have documented a pattern of broad claims covering conditions such as:
- Autism
- Neurological disorders
- Musculoskeletal injuries
- Chronic pain
- Degenerative diseases
Pew Charitable Trusts report on harms linked to unapproved stem cell interventions:
https://www.pew.org/-/media/assets/2021/05/harms-linked-to-unapproved-stem-cell-interventions-highlight-need-for-greater-fda-enforcement.pdf
While this data focuses largely on the United States, it illustrates the potential consequences when commercial stem cell offerings expand faster than regulatory clarity.
Introducing Wellbeing International Foundation Ltd
Among UK-registered entities operating within the broader wellbeing and regenerative medicine space is Wellbeing International Foundation Ltd, a company listed in Bermuda as a private limited company.
At the time of writing, there is no publicly available MHRA authorisation listing a stem cell product approved under that company name for general commercial use.
That does not automatically imply wrongdoing.
However, it raises key investigative questions:
- Are any therapies associated with the company classified as ATMPs?
- If so, have they received MHRA approval?
- If not, under what regulatory pathway are services being provided?
- Are treatments part of registered clinical trials?
- Are patients clearly informed of regulatory status before consenting?
These are regulatory classification questions — not allegations.
But they matter.
The Hospital Exemption Question
Under UK law, a hospital exemption allows certain unlicensed ATMPs to be used in specific circumstances under strict conditions.
However, this exemption:
- Must be authorised
- Must be non-routine
- Must be manufactured to specific standards
- Cannot be broadly marketed to the general public
The distinction between legitimate hospital exemption use and commercial marketing is critical.
MHRA position on clinical trials and exemptions:
https://www.gov.uk/government/publications/clinical-trials-regulations-enforcement-provisions/clinical-trials-regulations-enforcement-provisions
Marketing vs Medicine
A recurring issue across the regenerative medicine sector is the language used in advertising.
Regulators such as the Advertising Standards Authority (ASA) require that medical claims be supported by robust evidence.
Where treatments are described as “cutting-edge,” “innovative,” or “regenerative,” the regulatory classification becomes crucial:
- Is it a cosmetic procedure?
- Is it a medical service?
- Is it a medicinal product?
- Is it experimental?
Patients deserve clarity.
The Vulnerability Factor
Stem cell marketing frequently intersects with conditions that carry emotional weight:
- Autism
- Neurodegenerative disease
- Chronic pain
- Terminal illness
The Guardian recently reported concerns around companies promoting speculative future benefits of stem cell banking for children’s milk teeth — raising broader questions about evidence and consumer expectations.
Guardian investigation:
https://www.theguardian.com/science/2025/aug/21/healthcare-firms-misleading-milk-teeth-treatments-autism-diabetes-stem-cells
The common thread is hope.
Hope must never be monetised without evidence.
Key Investigative Questions for UK Patients
Anyone considering stem cell treatment in the UK should ask:
- Is this therapy MHRA-approved?
- Is it part of a registered clinical trial?
- Under what regulatory classification is it being offered?
- Has the Advertising Standards Authority reviewed any claims?
- What published, peer-reviewed evidence supports the treatment?
The absence of clear answers does not prove illegality.
But it should prompt caution.
Conclusion: A Sector in Need of Transparency
The UK’s regulatory framework for advanced therapies is strong — on paper.
But enforcement depends on classification, transparency, and proactive oversight.
Entities operating in the regenerative medicine and stem cell sector — including Wellbeing International Foundation Ltd — may find themselves under increasing scrutiny as policymakers and regulators assess whether current safeguards are sufficient.
This is not an accusation.
It is an investigation.
And in the field of regenerative medicine, clarity is not optional.
It is essential.
Recent Posts