QUESTIONS RAISED ABOUT A GERMAN LABORATORY LINKED TO HIGH-COST CELL-BASED TREATMENTS
An examination of recorded conversations, published claims, and regulatory concerns
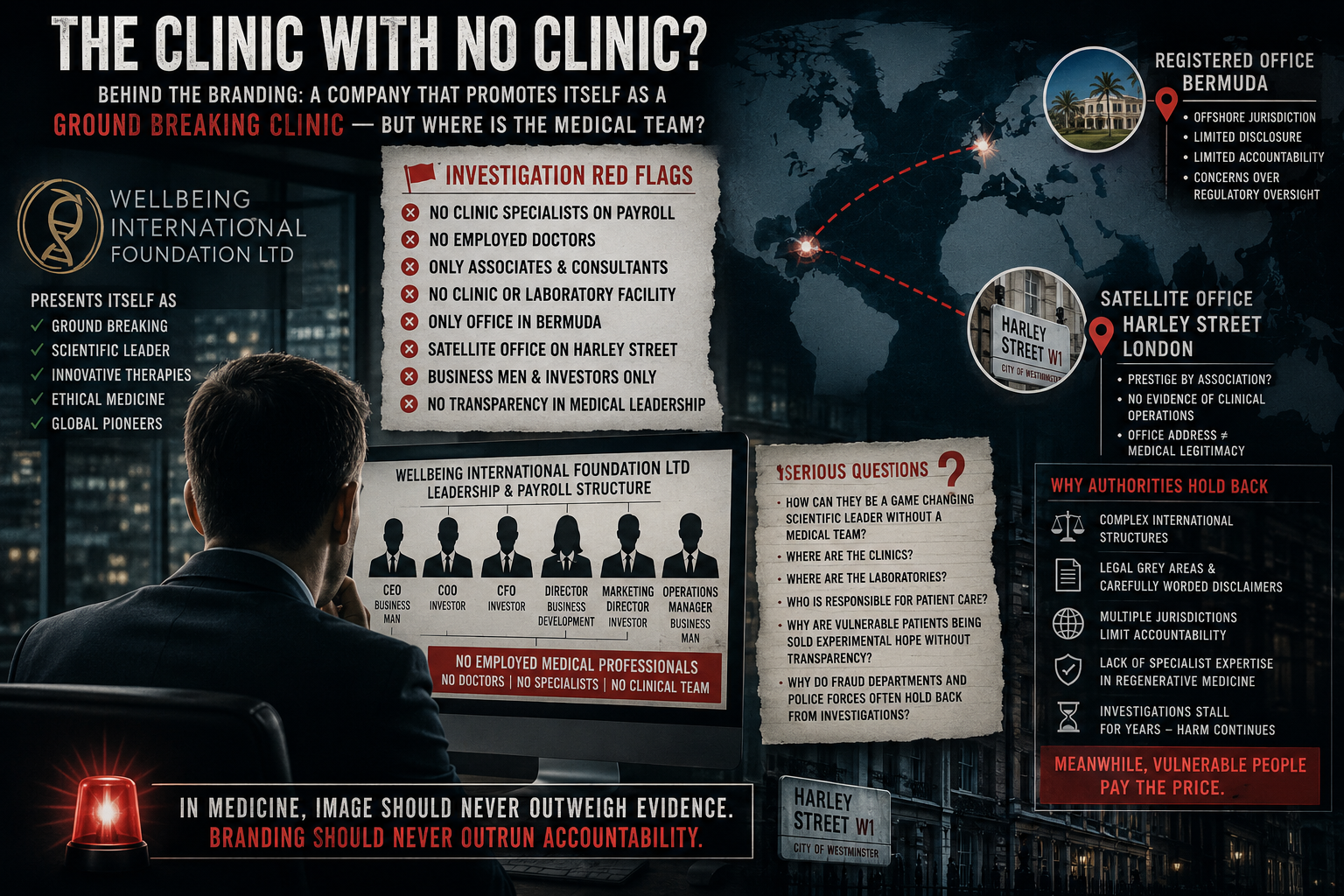
An independent investigation has raised serious questions about the role of a German laboratory, Ad Lentus, in the processing of blood-based therapies marketed to patients by Wellbeing International Foundation Ltd (WIF).
The concerns arise from a combination of published claims on Ad Lentus’s own website, recorded conversations involving its representatives, and expert opinion regarding laboratory capability and regulatory compliance.
Public claims of cancer treatment
On its publicly accessible website, Ad Lentus describes work involving autologous dendritic cell vaccines, which it characterises as a form of immunotherapy for the treatment of cancer. The website states that this approach falls within the category of Advanced Therapy Medicinal Products (ATMPs) and involves activating a patient’s immune system using cells derived from their own blood.
Such claims are significant. Under UK and EU law, the advertising and provision of cancer treatments are subject to strict regulation, and therapies described as ATMPs are normally required to undergo extensive authorisation, clinical trials, and regulatory approval.
Experts consulted for this investigation noted that publicly claiming to provide cancer treatments based on cell-derived products would typically require demonstrable compliance with Good Manufacturing Practice (GMP) standards and oversight by medicines regulators.
Recorded conversation with laboratory representative
As part of the investigation, an undercover operative (UC) participated in a recorded Zoom call involving Jörn Steiger, who is presented on the Ad Lentus website as a member of its team, and Andrew Chancellor, who has previously been recorded promoting high-cost treatments on behalf of WIF.
The recordings and transcripts have been preserved as exhibits and, according to the investigators, copies have been provided to both the US Food and Drug Administration (FDA) and the UK Serious Fraud Office (SFO).
During the call, Chancellor describes a long-standing working relationship between himself, Steven Ray, and Steiger, stating that they have collaborated since around 2010–2011 on various cell-based protocols.
Steiger confirms that Ad Lentus carries out laboratory work “according to GMP standards” and says the laboratory performs protocols designed by WIF.
Description of laboratory processes
In the recorded exchange, Steiger explains that blood-derived cells are isolated and then subjected to a form of stress intended to stimulate the release of cytokines and microvesicles. He states that no drugs or chemical additives are introduced and that the final infused product consists solely of material derived from the patient.
Steiger characterises this process as accelerating what cells would “normally do under natural conditions,” producing what he describes as “help factors” released in response to stress.
However, independent experts later told the investigation that the description provided raises questions about whether the laboratory processes described are sufficient to produce a consistent, clinically meaningful therapeutic product — particularly one claimed to have effects on cancer, inflammation, or tissue repair.
Questions about laboratory capability
Investigators say they have received information from a person familiar with the Ad Lentus laboratory setup suggesting that the facility does not possess the specialist equipment required to carry out the advanced cell processing described. That individual has not been identified due to safety and professional concerns.
While this claim cannot be independently verified at this stage, experts consulted for the investigation noted that producing ATMP-class therapies typically requires highly specialised infrastructure, validated processes, and regulatory oversight.
One scientist described the gap between how such therapies are discussed in marketing materials and what is realistically achievable in many private laboratories as “a recurring issue in this sector.”
Wider concerns about blood handling and oversight
The recorded conversations also raise questions about how patient blood is collected, transported, processed, and returned.
In earlier undercover recordings involving WIF representatives, patients were told that blood could be collected at home, shipped internationally, processed, and later re-infused — sometimes without involvement from the patient’s own doctor.
Investigators say they have identified the company used to transport blood to Germany and are examining whether appropriate licences and authorisations were in place. These issues will be examined in further reporting.
Context: a growing and controversial industry
Cell-based and “regenerative” therapies have become a rapidly expanding global industry, often operating in regulatory grey areas. Scientists and regulators have repeatedly warned that complex biological terminology can be used to give an impression of legitimacy without corresponding clinical evidence.
Experts consulted for this investigation emphasised that claims relating to cancer treatment, immune system “education,” or disease modification require especially robust evidence.
Public interest
This article is based on recorded conversations, published website material, and expert analysis. It does not make findings of criminal liability.
The purpose of publication is to highlight practices and claims that independent experts say warrant closer scrutiny by regulators, particularly where laboratory-processed blood products are promoted for serious medical conditions.
Individuals and organisations named have been given, or will be given, an opportunity to respond as further reporting continues.
This article is published in the public interest.
Recent Posts