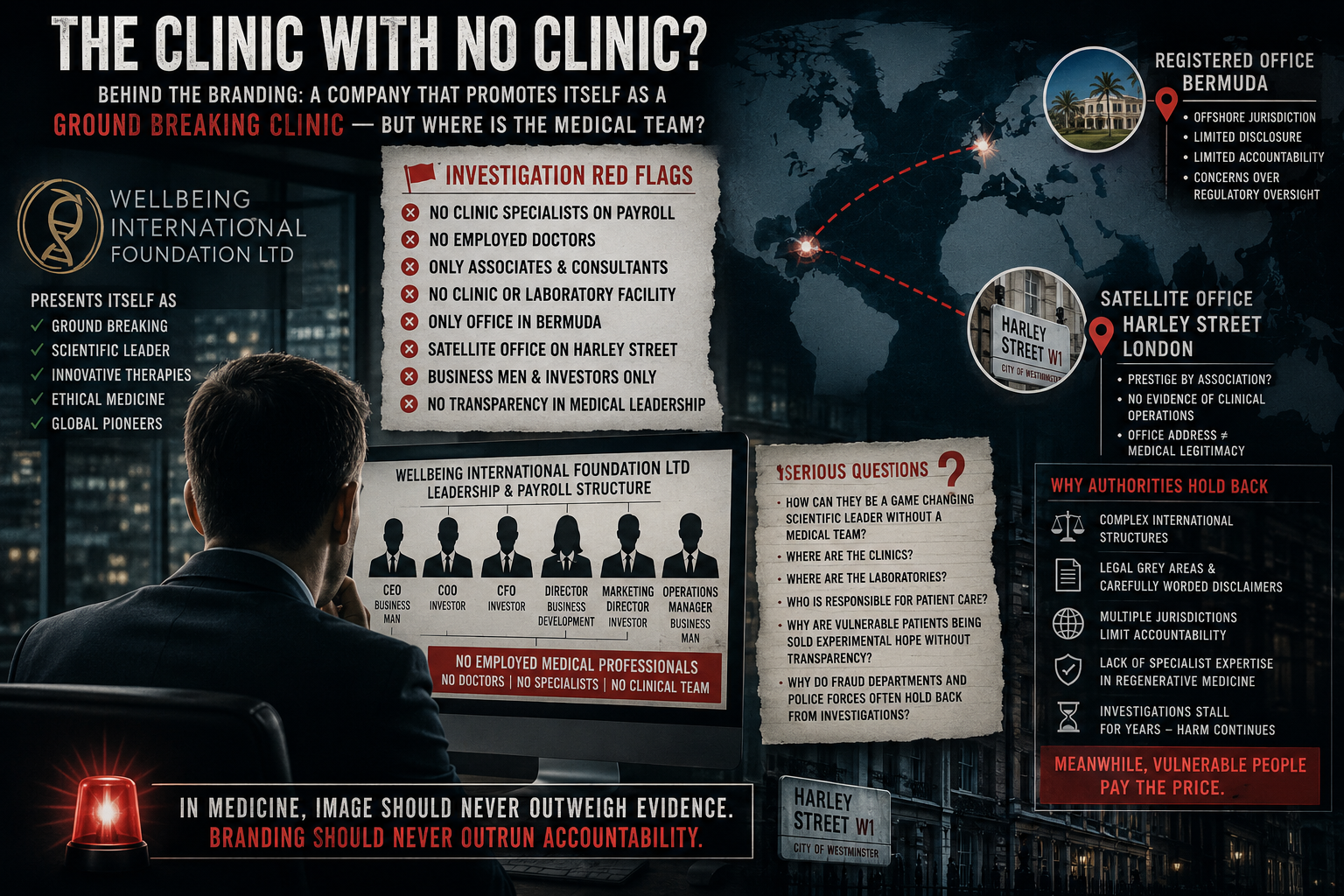
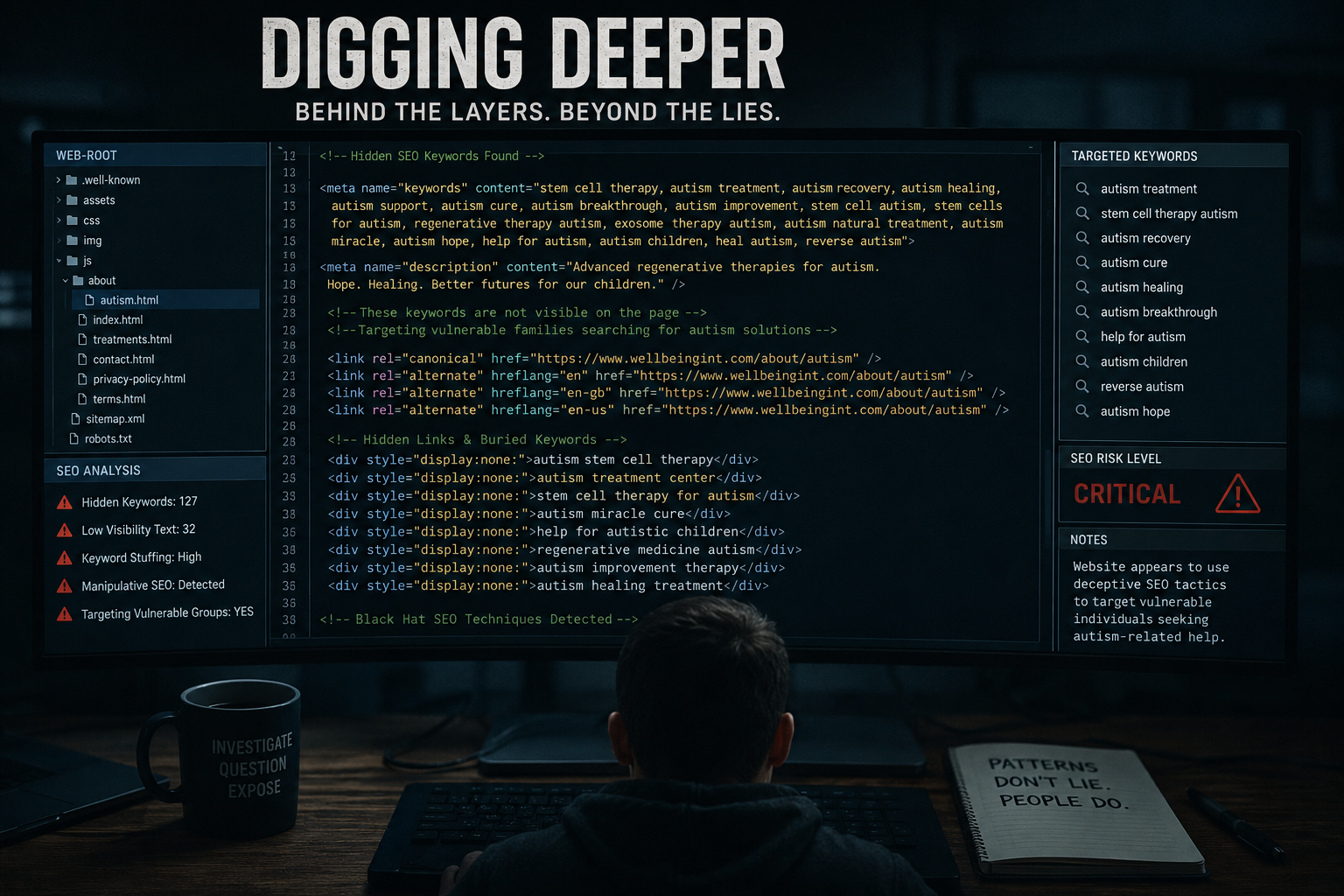
UNDERCOVER RECORDING RAISES SERIOUS CONCERNS ABOUT REMOTE SALES OF HIGH-COST STEM-CELL TREATMENTS
An analysis of a recorded telephone call with a non-medical sales representative
An undercover recording obtained as part of an independent investigation has raised serious concerns about how an expensive regenerative treatment was promoted to a prospective patient — without any medical consultation and by an individual who is not a doctor.
The recording captures a telephone conversation between an undercover operative (UC), based in the United States, and Andrew Chancellor (AC), who was acting on behalf of Wellbeing International Foundation Ltd.
During the call, AC discusses a proposed treatment costing $48,000, outlines how it would be delivered, and provides detailed medical claims — despite not being a qualified medical practitioner.
No consultation, no treating doctor involved
According to the recording, the entire interaction took place by telephone. At no point did AC advise the undercover operative to undergo a medical consultation with a doctor prior to treatment.
Nor did AC suggest involving the UC’s own physician.
Instead, AC states that the company could “find a doctor” to administer the treatment intravenously, distancing the procedure from the patient’s existing medical care.
In one key exchange, AC says:
“Well not your doctor, we can find a doctor for you, puts it into you intravenously.”
Experts later told this investigation that such a statement is significant, as it suggests awareness that a patient’s own doctor may be unwilling to administer the proposed procedure.
Medical claims made by a non-doctor
During the call, AC explains the treatment in highly technical terms, describing a process involving stem cells, laboratory manipulation, and the production of extracellular vesicles (EVs).
In the recording, AC claims that:
- stem cells would be taken from the patient’s blood
- the cells would be “grown” and placed under oxygen deprivation
- this process would generate billions of extracellular vesicles
- these vesicles would then be infused intravenously
- the vesicles would “seek out damage,” destroy dead cells, repair scar tissue, and improve inflammation
AC also suggests the treatment would reduce pain and improve function within “three to six weeks.”
These claims were made despite AC confirming neither a physical examination nor any form of medical assessment had taken place.
Logistics arranged without medical oversight
The recording further reveals that AC told the UC the treatment could be arranged almost anywhere, including the United States.
AC explains that:
- approximately 140–150ml of blood would be taken
- the blood would be shipped to a laboratory
- the processed material would later be infused intravenously
In a later email reviewed as part of the investigation, AC states that he could arrange for the blood to be taken at the patient’s home address.
Experts consulted for this investigation said that arranging blood collection and intravenous treatment in this manner, without documented medical oversight, raises significant safety and regulatory concerns.
Payment discussed before medical assessment
The conversation also includes a discussion of payment before any consultation or clinical review.
When asked about cost, AC states:
“The total cost to get all of this done and to have two treatments is $48,000.”
When asked about payment method, AC advises a direct bank transfer to a US bank account.
At no point in the call does AC discuss medical risks, alternative treatments, contraindications, or the need for informed consent.
Expert concerns
Independent experts who reviewed the transcript said the call raises multiple red flags.
They noted:
- medical claims being made by a non-doctor
- absence of a consultation or diagnostic process
- discouragement of involvement from the patient’s own physician
- high-pressure presentation of a costly intervention
- use of complex scientific language without supporting clinical evidence
One expert described this style of interaction as “deeply concerning,” particularly when aimed at individuals experiencing chronic pain or injury.
Public interest
This article is based on a verbatim transcript of an undercover telephone call, reviewed as part of an ongoing investigation into the marketing of unproven regenerative treatments.
The purpose of publication is to inform patients, families, and regulators about practices that independent experts say warrant close scrutiny — particularly where medical claims are made without clinical consultation or oversight.
The individuals and organisations referenced have been given an opportunity to respond prior to publication.
This article is published in the public interest.
Recent Posts