An investigation into claims made by a private stem-cell treatment provider
False Hope For Sale.
Inside a private medical office on London’s Harley Street, a patient describes a long list of health problems to Dr Stephen Ray. The consultation, later reviewed as part of an independent investigation, includes claims about advanced regenerative therapies that experts say are not supported by clinical evidence.
During the meeting, Dr Ray explains that his company, Wellbeing International Foundation Ltd (WIFL), offers treatments based on substances known as extracellular vesicles (EVs), which he describes as secretions produced by stem cells under stress. According to recordings of the consultation, blood would be taken from the patient, processed overseas, and re-administered as part of the therapy.
The proposed treatment, according to the recordings, was priced at £37,000.
Dr Ray also suggests that the treatment may have preventative effects against serious diseases. In the recording, he discusses neurodegenerative conditions such as Parkinson’s disease, stating that damage may begin years before symptoms appear and suggesting that early intervention could help the body repair itself.
However, multiple independent experts later told this investigation that such claims are not supported by clinical evidence.
The consultation itself was not arranged for treatment. The individual meeting Dr Ray was acting in an undercover capacity, recording the exchange as part of an investigation commissioned to examine the scientific and legal basis of the company’s claims.
Claims and evidence
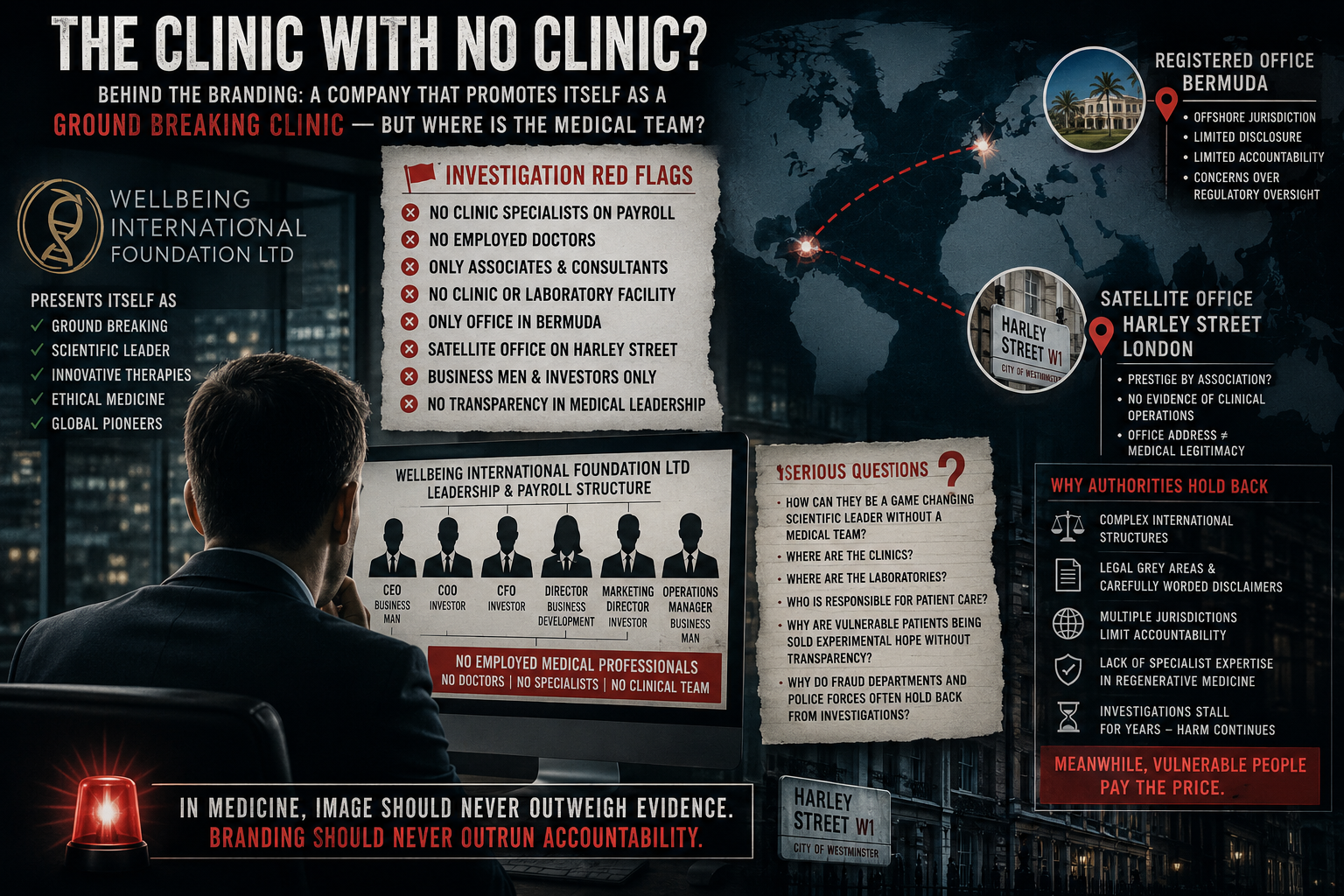
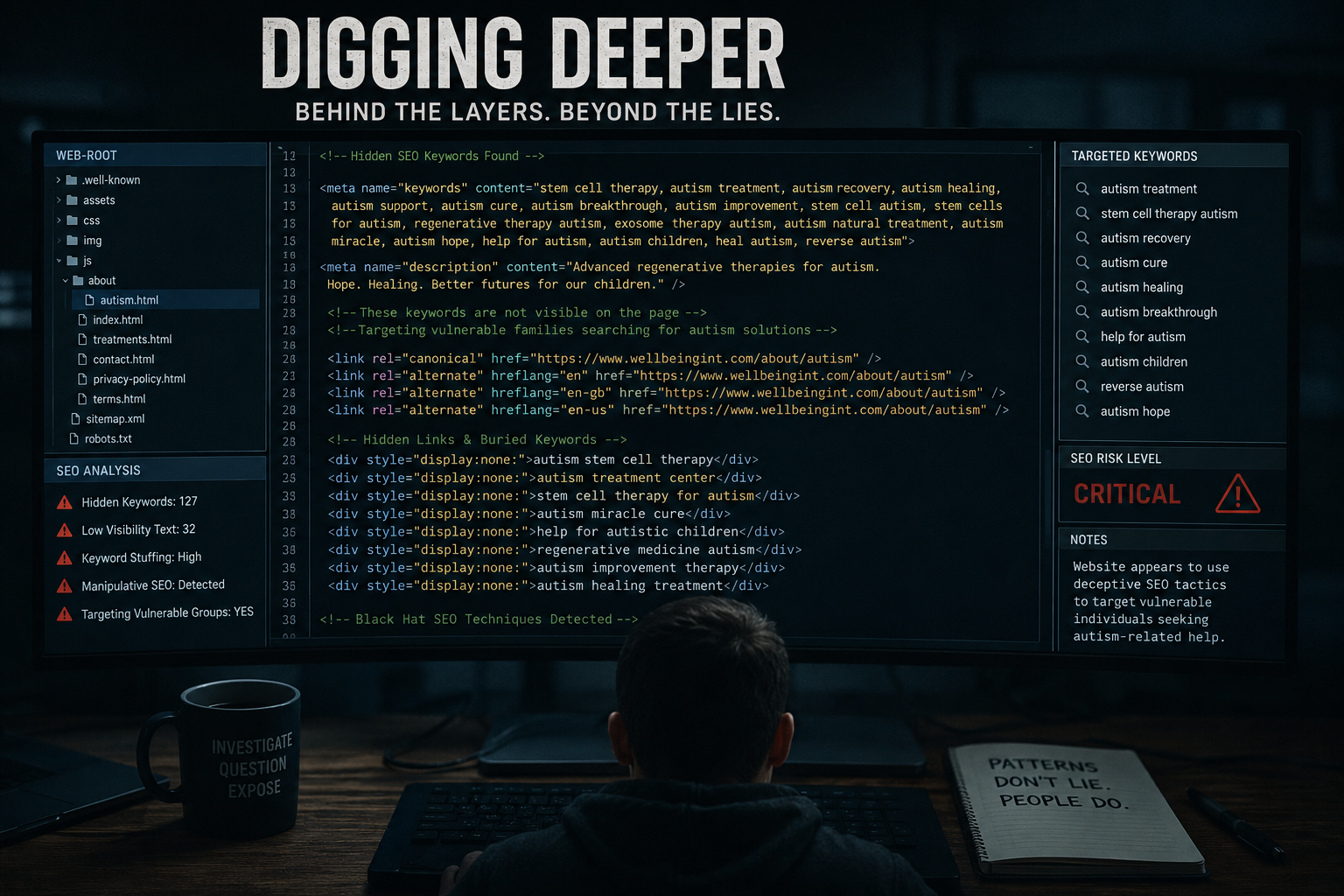
WIFL, which is registered in Bermuda, markets itself as a provider of regenerative medicine and promotes its services for a wide range of conditions, including sports injuries, chronic illness, and cancer. Its promotional materials include testimonials from athletes and individuals describing improvements in pain or mobility.
Central to the company’s marketing is its description of a “cell-free” therapy using extracellular vesicles rather than live stem cells. During the recorded consultation, Dr Ray states that these vesicles can stimulate repair, reduce inflammation, and potentially influence serious disease processes.
When asked in the recording about clinical evidence supporting the use of this therapy for neurodegenerative diseases, Dr Ray acknowledges that there is no primary research in humans, referring instead to a limited number of scientific papers.
Expert scientific assessment
An independent scientist specialising in stem-cell research, with a PhD in medical biotechnology, reviewed the recordings and the claims made during the consultation.
“There is legitimate preclinical research into extracellular vesicles,” the scientist said. “But moving from laboratory studies to clinical treatment in humans — especially for complex conditions like Parkinson’s disease — is an enormous leap. There is no robust clinical evidence to support what is being claimed here.”
The scientist said that using technical terminology could give the impression of scientific legitimacy without substantiating evidence.
“At best, based on what has been described, this appears to be an extremely expensive injection with no proven therapeutic benefit,” he said.
The expert also examined claims made during the consultation regarding Dr Ray’s academic background and research history.
“Several of the research activities described could not be substantiated through publicly available academic records,” he said. “A literature search shows limited published work, primarily involving behavioural studies in animals.”
He added that the laboratory processes described in the recordings did not align with established methods required to produce therapeutic extracellular vesicles.
“These are vulnerable people seeking answers,” the scientist said. “Selling unproven treatments at this price raises serious ethical concerns.”
Second expert opinion
A second independent expert, Dr Darius Widera, a specialist in stem-cell biology at the University of Reading, also reviewed the recordings.
“I listened carefully to the material,” he said. “The promises being made are not supported by evidence. There are no clinical trials demonstrating that this type of intervention can treat or prevent neurodegenerative disease.”
Dr Widera said claims about longevity or disease reversal belong “in the realm of science fiction.”
He acknowledged that some experimental research exists in orthopaedic contexts, such as knee injuries, but said results remain inconclusive.
“What is particularly concerning is the breadth of conditions being marketed,” he said. “From neurological disease to cancer — this goes far beyond what the science supports.”
Dr Widera also criticised the lack of transparency in how the therapies are described.
“The process is vague. There is no clear explanation of sourcing, processing, quality control, or regulatory oversight,” he said.
A growing industry and regulatory gaps
WIFL operates within a rapidly expanding global market for stem-cell-related therapies, estimated to be worth tens of billions of pounds. Growth in the sector has been driven by consumer demand, celebrity endorsements, and gaps in regulatory enforcement.
In the United States and Europe, regulators have issued repeated warnings about unproven stem-cell treatments, citing risks including infection, contamination, and the possibility that patients may delay effective medical care.
Dr Perry Wilson, a clinician and researcher at Yale University, has previously warned that clinical trial results for many stem-cell therapies are underwhelming.
“In conditions like arthritis, injecting plasma performs no better than saline in many trials,” he has said. “There are real risks, and the benefits are often overstated.”
Dr Widera believes regulatory uncertainty in the UK contributes to the persistence of such businesses.
“There is often confusion over responsibility,” he said. “In some European jurisdictions, similar practices would likely attract regulatory or criminal scrutiny.”
Legal concerns raised
A commissioned legal opinion reviewed the recorded material and marketing claims associated with WIFL. The opinion stated that the activities described raise serious concerns under UK consumer protection and fraud legislation, including the Fraud Act 2006.
The barrister noted that advertising unproven cancer treatments may also contravene the Cancer Act 1939.
“If the representations made to prospective patients are shown to be misleading and unsupported, they may amount to fraudulent misrepresentation,” the opinion stated.
The advice further noted that regulatory classification of certain treatments as “cell-free” may allow providers to avoid stricter oversight, despite making medical claims.
Public interest
Experts consulted for this investigation emphasised that the issue extends beyond a single company.
“What is being sold is hope,” one scientist said. “But hope without evidence can be harmful — financially, emotionally, and medically.”
This investigation is based on recorded consultations, expert analysis, and legal opinion. Individuals and organisations referenced were given an opportunity to respond prior to publication.
This article is published in the public interest to inform patients, families, and regulators about practices that independent experts say warrant closer scrutiny.
Recent Posts