PART TWO: THE CANCER QUESTION — WHAT WAS SAID IN THE RECORDED MEETING
Undercover footage raises further questions about how regenerative therapies are discussed with vulnerable patients

This is Part Two of an ongoing investigation into Wellbeing International Foundation and the individuals connected to its promotion of regenerative treatments. Part One examined recorded admissions regarding qualifications and professional roles.
During the same face-to-face meeting captured on video, undercover investigators questioned Stephen Ray — described as Chief Scientist — about claims surrounding the potential use of treatments in individuals suffering from serious illnesses, including cancer.
The exchange has prompted renewed discussion about how emerging therapies are communicated to patients and where legal boundaries may lie under UK law.
WHAT WAS SAID ON CAMERA
Throughout the conversation, Ray emphasised that he does not perform medical procedures himself and described his role as explaining scientific concepts rather than delivering clinical care.
When questioned about the effectiveness of treatments, he stated:
“I can speak to the science… I don’t do the medical treatment because I’m not a doctor.”
Investigators challenged him directly with concerns raised by external medical experts, suggesting that authorities in multiple countries were examining the company’s activities.
Ray responded by defending the underlying science and stating his belief that the approach works, while maintaining that patients ultimately decide whether to proceed.
The recording does not show Ray presenting himself as a licensed medical practitioner.
HOW THE PROCESS WAS DESCRIBED
In the discussion, Ray outlined a process involving:
- blood taken from the patient
- laboratory handling overseas
- re-introduction of material into the body at a later date
He described the therapy as involving “secretions that come off the stem cells,” rather than direct stem-cell transplantation.
When asked whether he believed the procedure treats people, Ray answered:
“Yes it does… based on the research we’ve actually done.”
THE LEGAL CONTEXT: THE CANCER ACT 1939
The recorded conversation included references to serious illnesses, prompting questions about the legal framework governing medical advertising in the United Kingdom.
Under the Cancer Act 1939, it is a criminal offence to advertise or promote treatments claiming to cure cancer unless specific exemptions apply. The law was introduced to protect vulnerable individuals from unproven or misleading medical claims.
This investigation does not allege that Ray personally breached the Act. However, legal experts say that any discussion linking treatments to serious diseases must be handled with extreme care, particularly when presented to prospective patients.
One healthcare regulatory analyst told this publication:
“The key issue is not just what is said — but how it might be interpreted by someone seeking hope for a serious condition.”
SCIENCE, SALES — OR SOMETHING IN BETWEEN?
During the meeting, Ray positioned himself as a communicator of scientific theory rather than a clinician.
He stated that his role was to explain what the treatment could and could not do, while acknowledging that final decisions rested with patients.
Critics argue that the distinction between scientific explanation and medical recommendation can become blurred when discussions take place in prestigious medical environments such as Harley Street — a location widely associated with high-level clinical expertise.
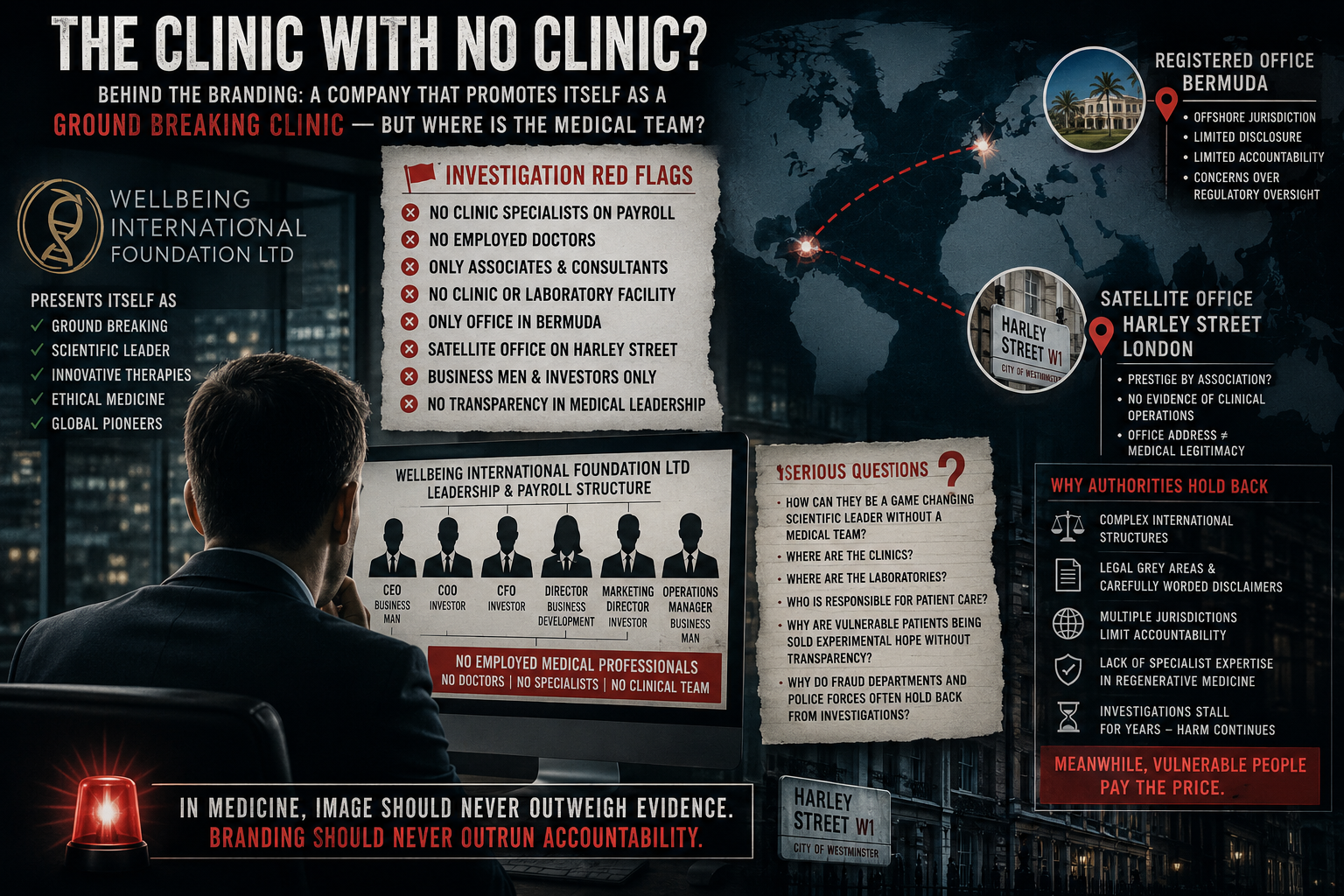
OFFSHORE STRUCTURES AND GOVERNANCE QUESTIONS
When asked why the organisation connected to the treatments was registered in Bermuda, Ray said:
“Max has got various business interests over there… I just deal with the science.”
The comment adds to ongoing questions about corporate structure and oversight raised in Part One of this investigation, which examined the business histories of key figures linked to the organisation.
WHY THIS RECORDING MATTERS
The significance of the footage lies not in a single statement, but in what it reveals about the wider ecosystem surrounding emerging regenerative therapies:
- non-medical scientific figures interacting with prospective clients
- complex international corporate structures
- treatments described in scientific rather than clinical terms
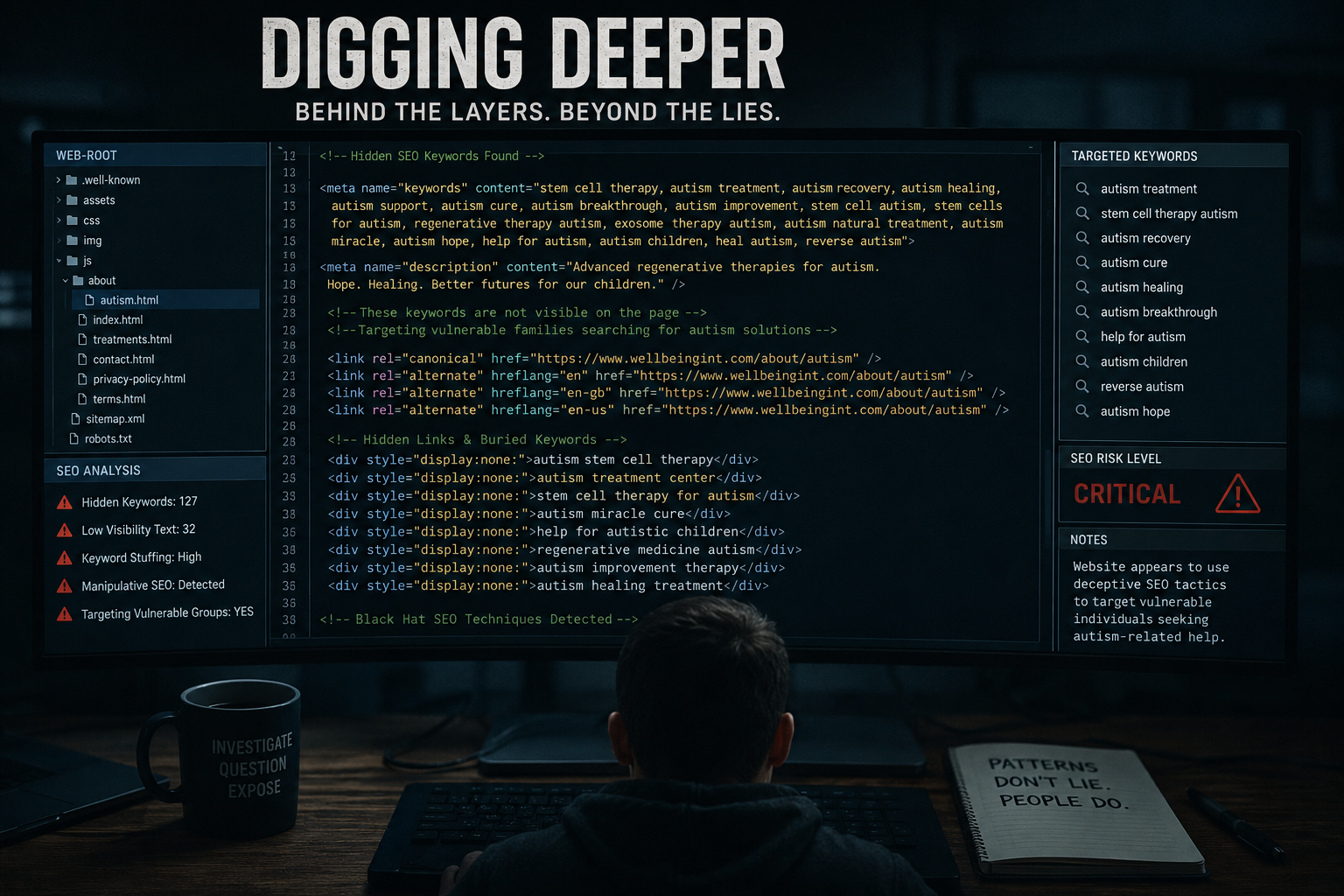
Supporters argue that innovative therapies often sit on the edge of traditional regulation. Critics counter that vulnerable individuals may struggle to distinguish between scientific discussion and medical advice.
THE INVESTIGATION CONTINUES
Wellbeing International Foundation has not responded to detailed questions at the time of publication.
This investigation will continue to examine how emerging therapies are presented to the public, how corporate structures influence patient access, and how regulators across multiple jurisdictions respond to rapidly evolving claims.
Recent Posts