HOPE, SCIENCE — AND HARD QUESTIONS
Undercover exchange sheds new light on cancer-linked treatment claims

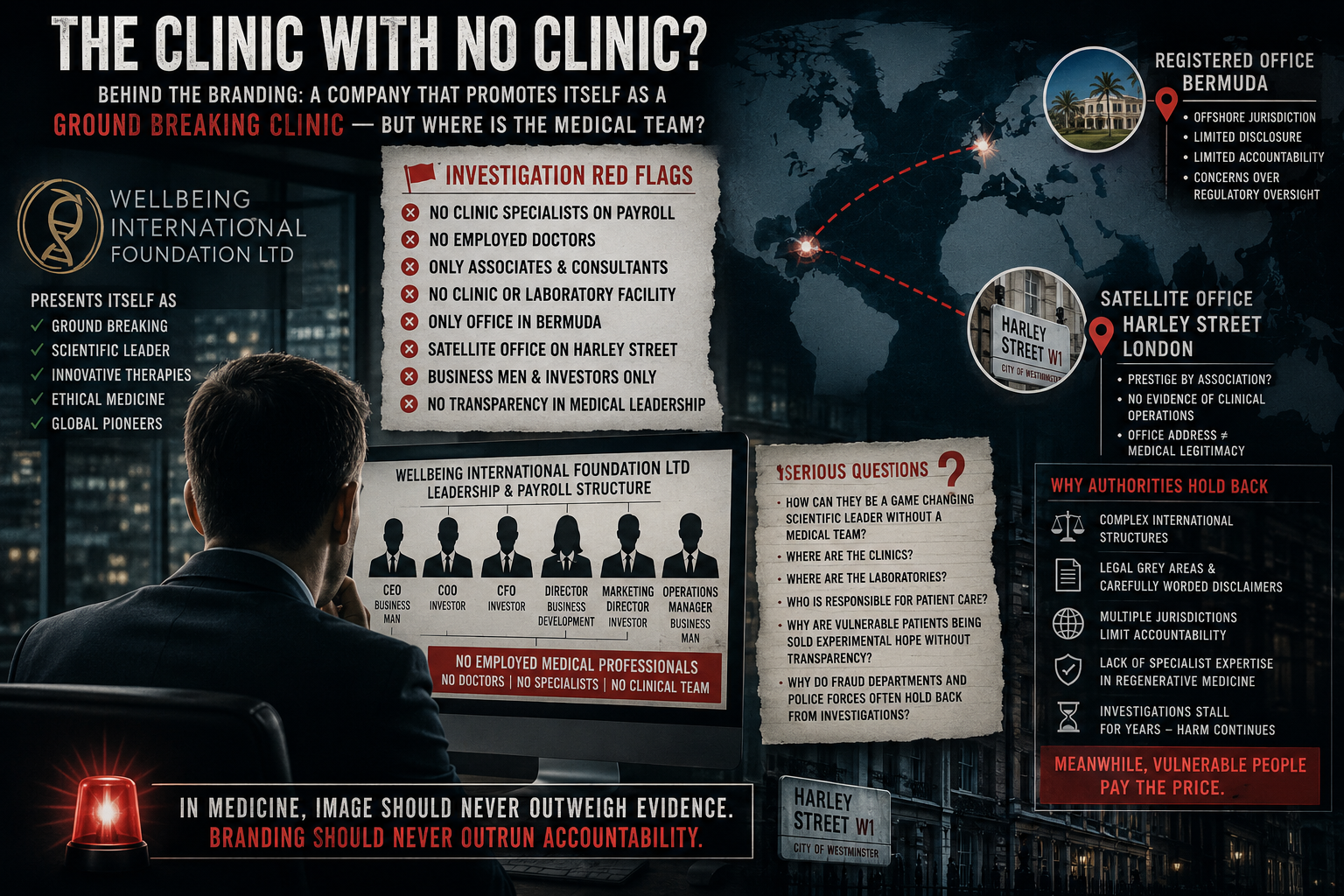
A covertly recorded conversation with a key figure associated with Wellbeing International has opened a new chapter in an ongoing investigation into the marketing of experimental regenerative therapies.
In the meeting, the individual — who holds a PhD but is not a medically licensed doctor — attempted to distance the organisation’s work from traditional stem-cell treatments, describing instead a focus on dendritic cells, extracellular vesicles and other forms of immunotherapy.
When questioned about cancer-related claims, he emphasised that the therapies were not positioned as replacements for surgery, chemotherapy or radiotherapy.
“This isn’t an alternative to conventional therapy,” he said, framing the approach as a potential complement to established medical care.
Yet the exchange revealed tensions between scientific language and patient expectations — particularly when the undercover journalist pressed for specific evidence supporting the claims.
Literature — But Which Evidence?
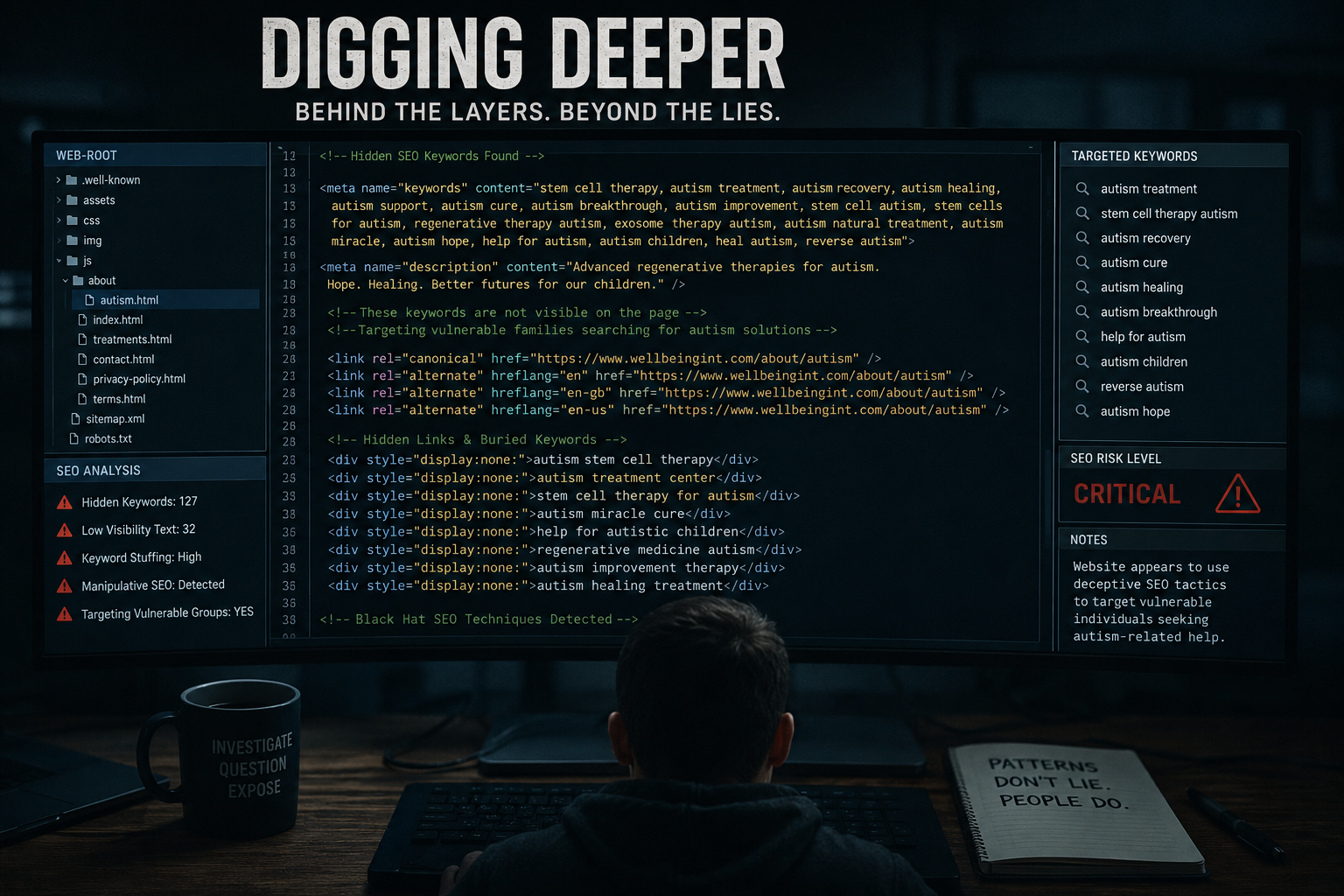
During the discussion, the interviewee referred repeatedly to a “large body of literature” surrounding immunotherapy and exosomes.
No individual studies were cited at the time, and the reporter questioned how patients were meant to distinguish between early-stage research and clinically validated treatments.
Observers of the private regenerative medicine sector have long argued that scientific terminology can be difficult for non-specialists to interpret — especially when individuals are seeking options during serious illness.
Concerns Over Patient Screening
One of the most contentious moments arose when the reporter raised allegations that some prospective clients had not been asked to provide full medical records before discussions about treatment.
The interviewee rejected any suggestion that documentation would be refused, stating that records were welcome where available.
However, the journalist maintained that previous conversations with patients suggested otherwise — highlighting what critics say may be inconsistencies in how clinics assess medical suitability.
The Cost Question
Another striking exchange focused on pricing.
The individual acknowledged having little involvement in financial arrangements but estimated that packages could reach around 50,000 US dollars.
The reporter challenged whether a patient-facing figure should remain unaware of costs that high — particularly when treatments are marketed to individuals dealing with serious diagnoses.
“I can only speak to the science,” he responded.
Legal Grey Areas
The conversation took a legal turn when the undercover journalist referenced the UK’s Cancer Act 1939, legislation that restricts certain types of cancer-related advertising.
The interviewee indicated he was not familiar with the law and suggested that helping patients alongside conventional treatment was different from claiming to treat cancer directly.
That distinction reflects a broader regulatory debate currently unfolding in multiple countries, where emerging therapies often sit in a space between research and commercial practice.
Pressure Behind the Scenes
As the discussion grew more direct, the reporter suggested that senior figures connected to the organisation remained largely absent from patient consultations, leaving others to represent the science publicly.
The interviewee confirmed long-standing professional relationships but declined to elaborate on internal roles or decision-making.
When asked whether he would consider assisting authorities or acting as a whistle-blower, he answered simply: “No.”
A Growing Debate
The exchange ended with both sides firmly holding their ground.
The interviewee argued that extracellular vesicle research was expanding rapidly, citing thousands of publications, while the undercover journalist questioned whether commercial applications had moved ahead of clinical consensus.
The encounter reflects a wider global argument: when does experimental medicine become premature marketing — and who decides where that line sits?
CONTINUING COVERAGE
Further recordings, set to be released in the coming weeks, explore additional questions surrounding patient communication, oversight and the business structures behind the treatments.
Recent Posts