STEM CELL GOLD RUSH: INSIDE THE UK’S BOOMING — AND UNPROVEN — TREATMENT INDUSTRY
Hope, For a Price

For tens of thousands of pounds, patients across the UK are being offered stem cell and exosome-based treatments promising relief from chronic pain, degenerative conditions, and even neurological disorders.
The language is persuasive. The science sounds cutting-edge. The settings — often prestigious London addresses — suggest credibility.
But behind the marketing, a growing number of experts are raising serious concerns.
A Rapidly Expanding Private Market
Recent reporting has indicated that more than 70 private providers in London alone offer stem cell treatments for joint pain. These procedures typically involve extracting cells from a patient’s bone marrow or body fat before re-injecting them into affected areas.
Despite the scale of the industry, questions remain over the scientific foundation behind many of these treatments.
Leading orthopaedic voices have warned that, in many cases, there is no established evidence base — describing what critics call a “sham scientific legitimacy” driven by commercial interests.
Science vs Sales
Stem cell research is a legitimate and evolving field of medicine, with proven applications such as bone marrow transplants.
However, many of the treatments now marketed by private clinics — particularly those targeting joint pain, ageing, or neurological conditions — remain outside well-established clinical evidence.
Patients are often presented with:
- Emerging or experimental therapies
- Limited or inconclusive clinical data
- High-cost treatment packages outside mainstream healthcare pathways
Critics argue that the gap between scientific research and real-world application is increasingly being bridged not by evidence — but by marketing.
The Business of Treatment
Unlike NHS care, these treatments operate almost entirely within a private-pay model.
Patients typically:
- Discover clinics through online advertising or testimonials
- Enter consultation processes that resemble structured sales funnels
- Receive pricing structures that can exceed £20,000–£50,000
In many cases, access to treatment appears to depend less on strict clinical criteria and more on a patient’s ability to pay.
It raises a difficult question:
Where does medical treatment end — and commercial opportunity begin?
Growing Scrutiny and Police Interest
Concerns surrounding the industry are no longer confined to academic debate.
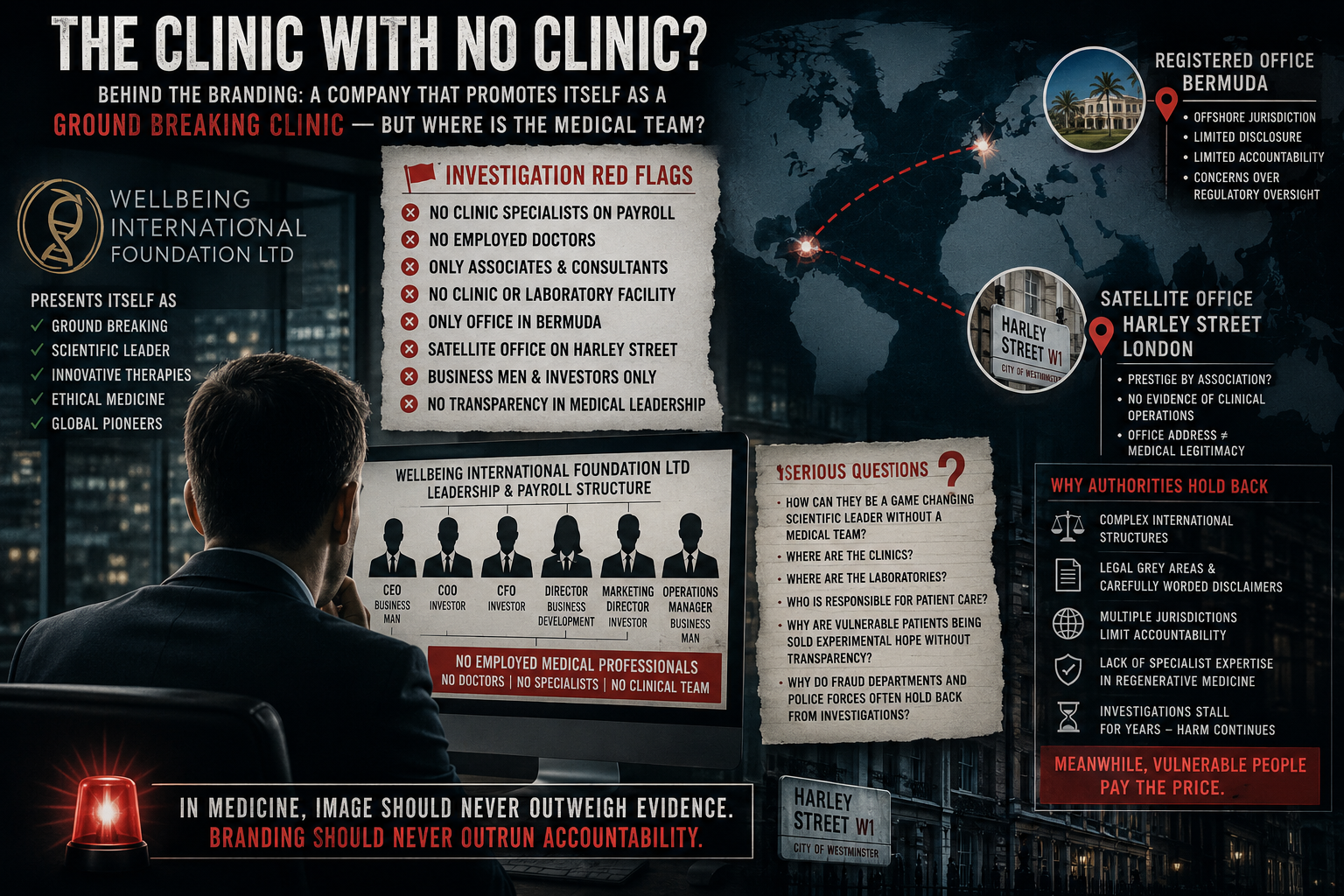
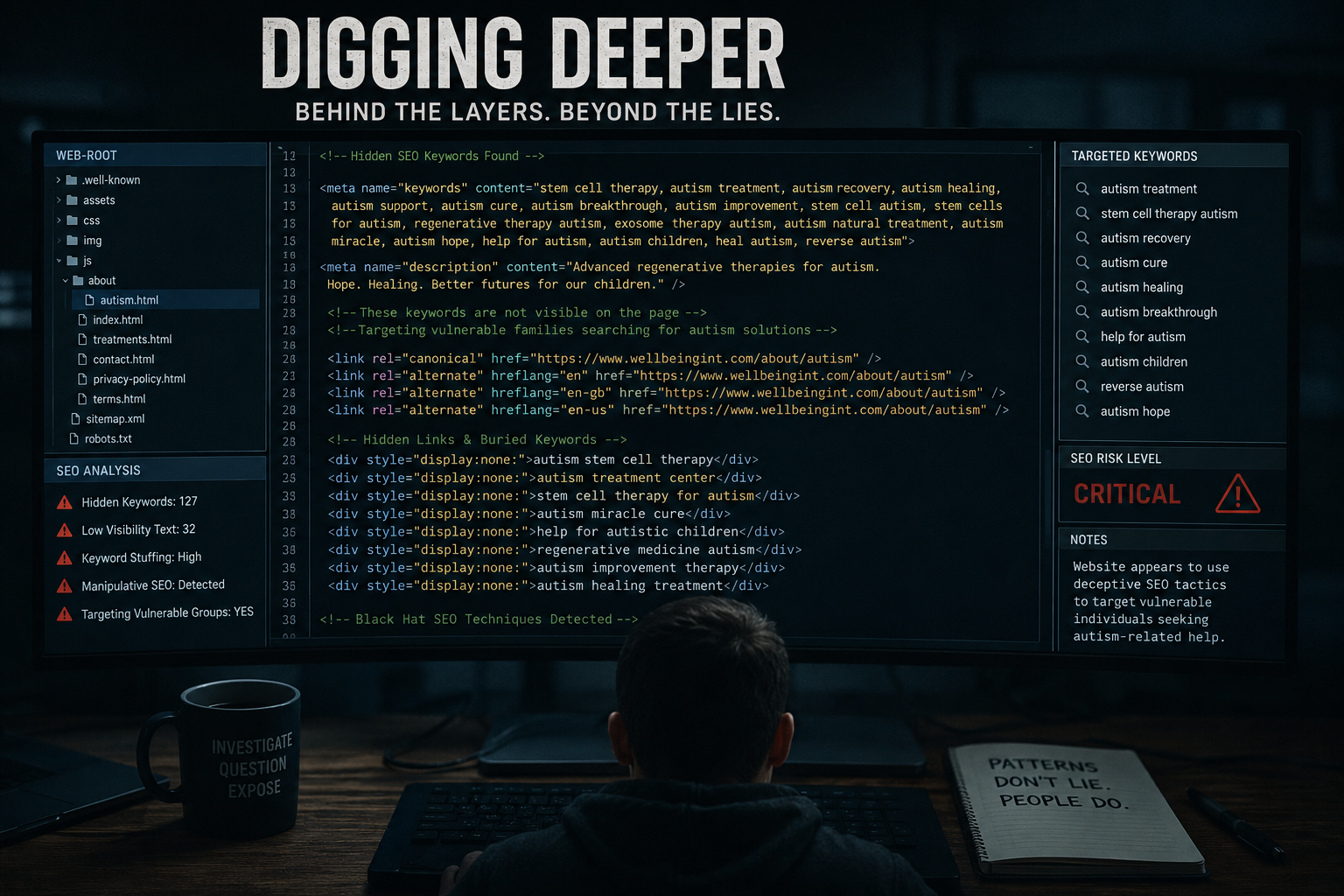
In April 2024, reports emerged of a Metropolitan Police investigation into a doctor offering stem cell treatments for children with autism — a particularly sensitive area where scientific evidence remains limited and heavily debated.
Previous coverage has also highlighted the unwarranted hype surrounding stem cell therapies for autism, especially in vulnerable patient groups.
While investigations remain ongoing, the case reflects increasing scrutiny of how far some providers may be extending treatment claims.
A Shadow of Scientific Controversy
Parts of the regenerative medicine sector trace their scientific roots to research environments that have themselves been subject to controversy.
Past investigations into research conduct at major European institutions — alongside high-profile medical scandals involving experimental procedures — have raised broader questions about oversight, accountability, and the translation of early-stage science into commercial treatment.
While such controversies are not directly linked to every clinic operating today, they highlight a recurring concern:
How scientific credibility is borrowed, interpreted, and ultimately sold.
Echoes of Past Healthcare Scandals
Observers have drawn cautious comparisons with previous healthcare controversies, where bold medical claims gained traction long before being scientifically validated.
The parallels are not exact — but the pattern is familiar:
- Rapid growth
- Strong consumer demand
- Limited regulatory clarity
- Innovation outpacing evidence
The Regulatory Gap
At the heart of the issue lies a widening gap between innovation and oversight.
Stem cell and regenerative therapies sit at the intersection of:
- Emerging science
- Private healthcare markets
- Global treatment access
Regulators face a difficult challenge — distinguishing genuine innovation from premature or overstated application, often after services have already reached patients.
A Market Ahead of Its Evidence
The promise of regenerative medicine is real.
But so too is the risk of overstatement.
As the UK’s private stem cell sector continues to expand, patients are increasingly navigating a marketplace where hope, science, and commercial ambition intersect — not always transparently.
Until clearer standards, stronger clinical evidence, and tighter oversight are established, one reality remains:
Patients may be paying for treatments that science has not yet fully proven — in an industry still defining its own boundaries.
Recent Posts