Wellbeing International Foundation Ltd: brand polish, bold claims, and the questions that still need answering
In the world of unproven regenerative medicine, the warning signs are rarely crude. They are polished. They are carefully worded. They speak of science, hope, recovery, and breakthroughs. And that is exactly why companies operating in this space deserve close scrutiny.

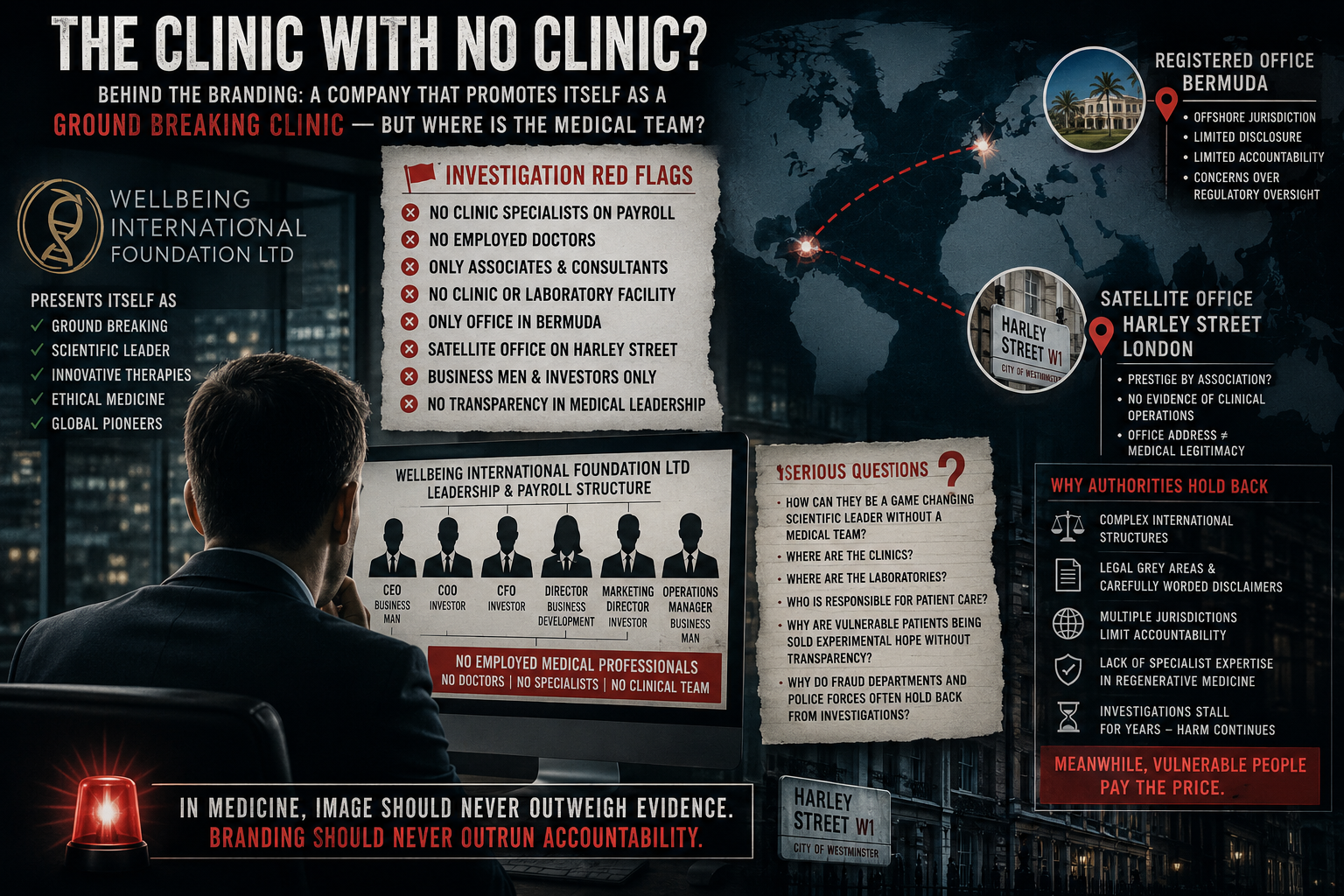
Wellbeing International Foundation presents itself as a cutting-edge biotech and regenerative therapy group based in Bermuda. On its website, it says it is backed by “30+ years of peer-reviewed research,” claims “more than 1,000 satisfied customers,” and promotes a “cell-free therapy” said to support injury recovery, immune resilience, longevity, and conditions including Parkinson’s and multiple sclerosis. It also says its process involves taking a patient’s blood, extracting stem cells and regenerative cells, “stressing” them to release exosomes and healing signals, and generating enough material for around 10 treatments. (Wellbeing International)
On its face, that is a compelling sales pitch. But it is also exactly the sort of pitch that should trigger hard questions.
The company’s public-facing material leans heavily on authority language: “peer-reviewed research,” “government regulated labs in the UK,” “scientific credibility,” and named executives and advisers. Its About page lists medical and scientific figures alongside commercial leadership, while also describing a global footprint across cities in the United States, Europe, Bermuda, Dubai, and New Zealand. (Wellbeing International)
At the same time, the promotional structure appears broader than a single corporate website. In addition to its main site, there is a separate testimonial-focused website dedicated almost entirely to positive stories and brand reinforcement, again promoting the same “cell-free therapy” narrative and broad wellness benefits. That second site says the business began treating selected clients in Switzerland in 2010 and is now headquartered in Hamilton, Bermuda. (Wellbeing International Foundation)
That matters, because in controversial medical sectors, reputation architecture is often just as important as the medicine itself. A main commercial site establishes legitimacy. A separate testimonial site amplifies social proof. A LinkedIn presence adds corporate gloss, describing the company as a biosciences business using “secretions from clients’ own pro-regenerative cells” and saying it was founded in Bermuda in 2015. (LinkedIn)
None of that proves wrongdoing. But it does raise an obvious question: is this science-led medicine, or marketing-led medicine dressed in scientific language?
Another point that deserves scrutiny is the name and structure around the brand. UK Companies House shows an active company called Wellbeing International R&D Limited, incorporated in January 2018, whose previous names were Game Mode Limited and later Biosystems & Technologies Limited before becoming Wellbeing International R&D Limited in June 2020. Companies House also lists Max Robert Lewinsohn as a current director. Companies House explicitly notes that it does not verify the accuracy of filed information.
That does not mean the Bermuda-facing Wellbeing International Foundation and the UK company are the same legal entity. But when a public-facing medical brand sits alongside a UK company with prior unrelated names, overlapping personnel, and “R&D” branding, investigators are right to ask whether the structure is straightforward or strategically layered.
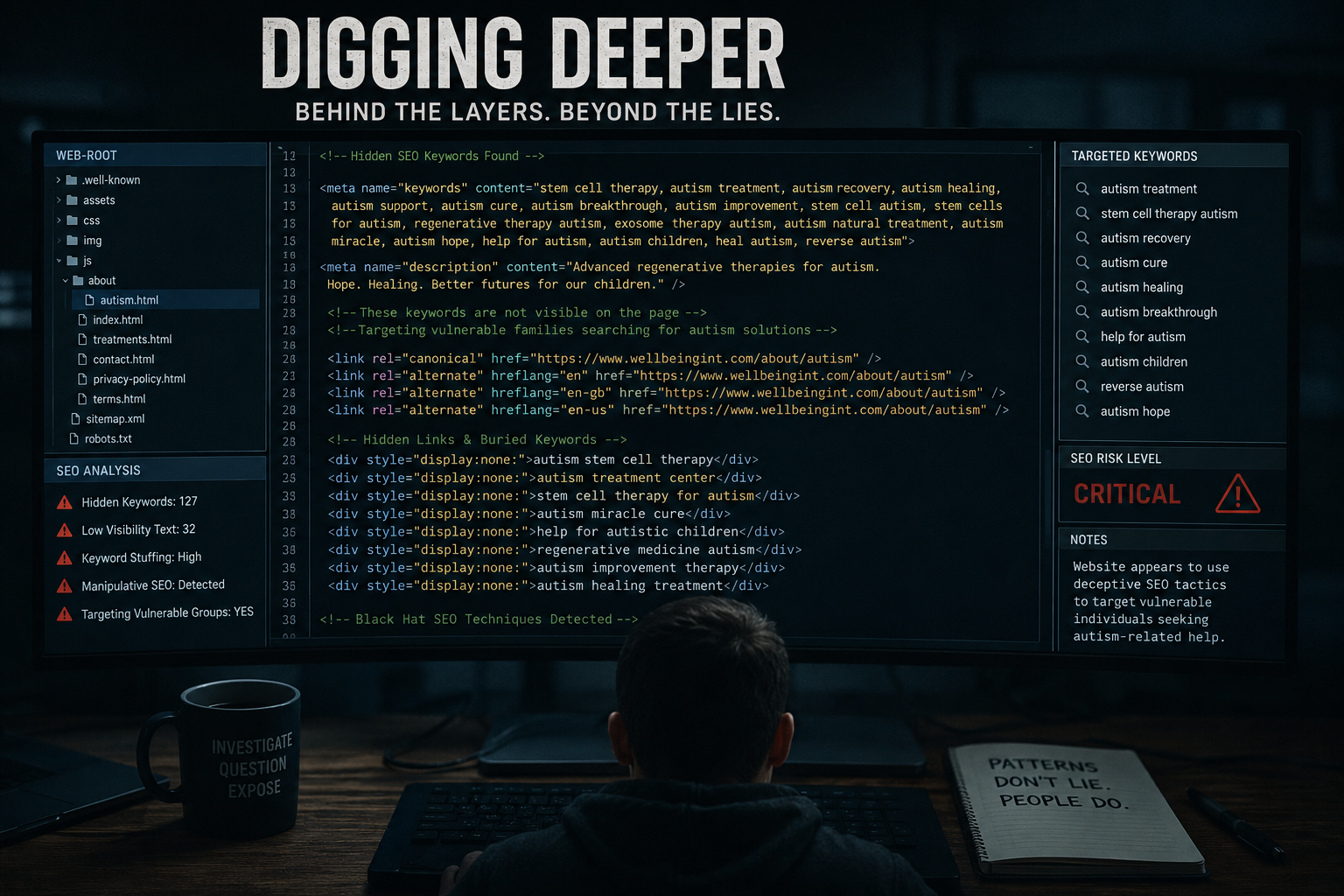
The treatment claims themselves also deserve careful examination. The company says its therapy can “accelerate healing,” can cross the blood-brain barrier, can help neurological conditions, and can be used both as a standalone treatment and alongside conventional medicine. It presents athlete endorsements and patient testimonials involving injury recovery, multiple sclerosis stability, chronic pain improvement, and broader vitality claims.
Testimonials, however, are not clinical proof. Celebrity endorsements are not clinical proof. A polished website is not clinical proof. And phrases like “30+ years of research” can create an impression of robust validation even when the actual public evidence available to a patient may be far less clear than the marketing suggests. That is the central problem in this sector: desperation is monetised through ambiguity.
The issue is not whether regenerative medicine has scientific promise. It does. The issue is whether vulnerable patients are being sold a level of certainty, effectiveness, and credibility that has not been independently demonstrated in a way that matches the boldness of the claims.
When a company markets hope in areas like neurodegeneration, anti-ageing, chronic pain, and high-performance recovery, the burden should be on that company to show clear evidence, clear regulation, clear accountability, and clear legal responsibility. Patients should not have to decode corporate structures, brand ecosystems, and testimonial funnels just to work out who they are really dealing with.
For investigators, the questions now are simple:
- Who is the true operating entity behind the treatment?
- What independent clinical evidence supports the claims being made to the public?
- How are patients marketed to, qualified, charged, and followed up?
- And why does the brand ecosystem appear to rely so heavily on promotional storytelling and reputation-building assets?
Until those questions are answered clearly, skepticism is not cynicism. It is consumer protection.
Recent Posts